The widely circulated notion of "hair porosity" and the popular at-home tests designed to determine it are fundamentally flawed, according to scientific research. While haircare advice often frames product recommendations around concepts of "low" or "high porosity," the common float and drop tests do not accurately measure the hair’s internal absorption capabilities. Instead, these tests primarily indicate the surface condition of hair, particularly the presence and integrity of its outermost protective layers, rather than its intrinsic ability to absorb moisture. This scientific discrepancy highlights a significant gap between popular beauty wisdom and dermatological and trichological understanding, prompting a re-evaluation of how consumers and professionals approach hair health assessments.
The Rise of Hair Porosity in Beauty Culture
The concept of hair porosity has gained immense traction within the beauty industry, especially among communities focused on natural and textured hair. It purports to describe how easily moisture can penetrate and be retained by the hair strand. Generally, "low porosity" hair is said to have tightly bound cuticles, making it difficult for water and products to enter, but once inside, moisture is well-retained. "High porosity" hair, conversely, is characterized by open or raised cuticles, allowing easy absorption but also rapid loss of moisture. "Medium porosity" is often considered the ideal balance. This framework has led to a cascade of product formulations and regimen suggestions tailored to each porosity type, from lightweight humectants for low porosity to heavier butters and oils for high porosity.
The allure of these classifications stems from the promise of personalized haircare, empowering individuals to select products best suited to their unique hair needs. To aid in this self-diagnosis, several DIY tests have become ubiquitous on social media platforms and beauty blogs. The two most prominent are the "Float Test" and the "Drop Test," both presented as simple, at-home methods to determine one’s hair porosity. However, a deeper look into hair science reveals that these tests provide misleading information, often leading to correct product choices purely by coincidence rather than accurate diagnosis.

Debunking the "Waterproof Hair" Myth
A foundational myth underpinning the flawed porosity tests is the idea that healthy, undamaged hair is inherently waterproof. This misconception suggests that the hair cuticle, when intact, forms an impenetrable barrier, sealing out water and other external elements. Conditioners are often marketed as mimicking this sealing function for damaged hair. However, scientific evidence directly contradicts this notion. Human hair, even in its most pristine, undamaged state, is far from waterproof.
Hair is a hygroscopic material, meaning it readily absorbs moisture from its environment. Research shows that undamaged hair can absorb a substantial amount of water—up to 30% of its dry weight—within minutes. This absorption is dynamic and responsive to environmental humidity. For instance, studies by Robbins C.R. in "Chemical and Physical Behavior of Human Hair" demonstrate a clear relationship between relative humidity and the weight of water absorbed by hair:
| Relative humidity (%) | Weight of water absorbed (%) |
|---|---|
| 0 | 0 |
| 8 | 3.9 |
| 40 | 10.2 |
| 63 | 14.8 |
| 86 | 22.6 |
| 100 | 31.2 |
Source: Robbins CR. Chemical and Physical Behavior of Human Hair. Springer Berlin Heidelberg 2012.
This data unequivocally illustrates that hair continuously exchanges moisture with its surroundings. The notion of a completely sealed, impermeable cuticle is therefore inaccurate. The hair cuticle, composed of overlapping scales, is often likened to shingles on a roof or the scales of a pinecone. While these scales provide a protective outer layer, they are not perfectly sealed. There are microscopic gaps and edges where water molecules, especially those in gaseous form (humidity), can penetrate and enter the hair’s inner cortex.
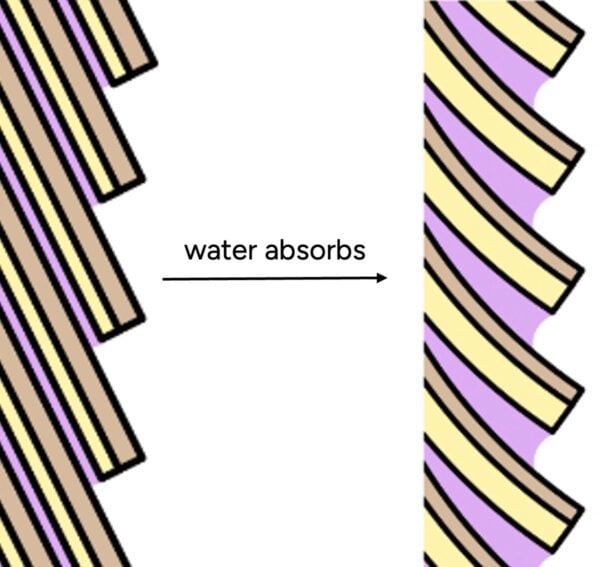
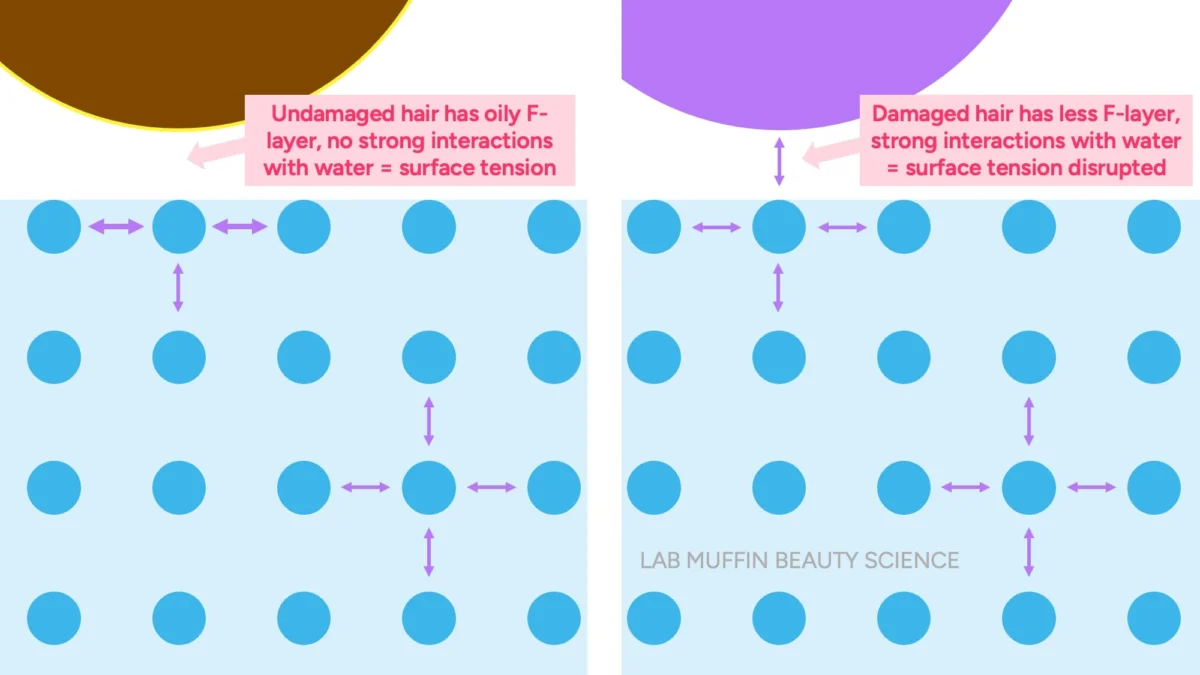
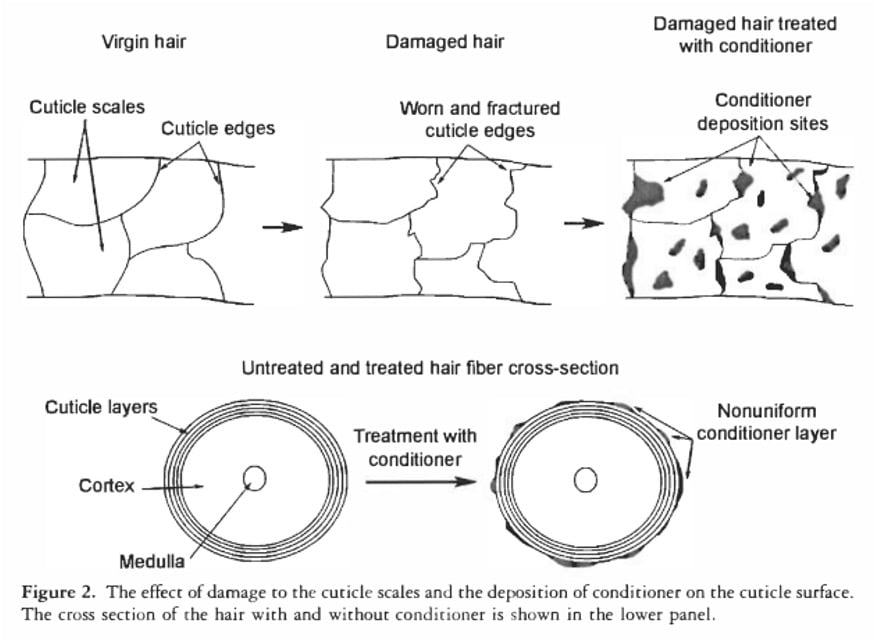
Furthermore, the natural conditioning layer of hair, known as the F-layer, resides only on the top surface of each cuticle scale. This means significant areas between scales remain exposed or only partially covered, providing ample pathways for water molecules to enter. Similarly, commercial conditioners, while effective at smoothing the hair’s surface and reducing friction, do not create a continuous, waterproof film. Microscopic analyses, such as those by La Torre C and Bhushan B (2006), reveal that conditioners deposit on the hair shaft in discrete "blobs" or patches, rather than a uniform, impermeable layer. While these microscopic deposits are sufficient to impart softness and reduce tangling by creating a smoother feel, individual water molecules are infinitesimally small and can easily navigate around and through these non-continuous barriers.
Debunking the At-Home Porosity Tests: The Role of Surface Tension
Given that hair is not waterproof and readily absorbs moisture, the underlying premises of the float and drop tests crumble under scientific scrutiny. These tests, rather than measuring the internal porosity or absorption capacity, are actually indicators of how water interacts with the surface of the hair strand. The key scientific principle at play here is surface tension.
Surface tension is a property of liquids that results from the cohesive forces between molecules at the liquid’s surface. In water, individual water molecules are strongly attracted to each other through hydrogen bonds. Molecules within the bulk of the liquid are surrounded by other water molecules in all directions, creating a balanced network of attractions. However, water molecules at the surface have fewer neighbors above them (only air), leading to stronger inward pulls from the molecules below and sideways pulls from adjacent surface molecules. This imbalance creates a net inward force, causing the surface to behave like a stretched elastic "skin." This "skin" allows light objects, like insects or paperclips, to seemingly "float" on water, even if their density is greater than water, until the surface tension is disrupted. For example, a steel paperclip (eight times denser than water) can float until a detergent (a surfactant that reduces surface tension) is added or the surface is agitated. Both hair and a paperclip are dense enough to sink in water, but they only do so when this crucial surface tension is overcome.
The Float Test: A Misinterpretation of Surface Interaction
The Float Test typically involves placing a clean strand of hair into a glass of water and observing whether it sinks or floats. The popular interpretation suggests that hair that quickly sinks is "high porosity" because its open cuticles readily absorb water, increasing its density and causing it to descend. Conversely, hair that floats or sinks slowly is deemed "low porosity" due to tightly sealed cuticles preventing water absorption.
The scientific reality is different. When an undamaged hair strand, with its intact F-layer (a naturally hydrophobic, oily coating), is placed on water, the water molecules at the surface are unable to "hold hands" with the hair surface effectively. The hydrophobic nature of the F-layer resists interaction with water, allowing the water’s strong surface tension to remain largely undisturbed around the hair strand. This undisturbed surface tension acts as a supportive layer, enabling the hair to "float" on the surface, much like the paperclip example.
However, if the hair surface is damaged—due to chemical treatments (coloring, perming), heat styling, harsh brushing, or environmental exposure—the protective F-layer is compromised or removed. This exposes a more hydrophilic (water-loving) surface beneath. When this damaged, hydrophilic hair encounters water, the water molecules at the surface readily form hydrogen bonds with the hair. This interaction disrupts the cohesive network of water molecules at the surface, effectively weakening or breaking the "skin" of surface tension. With the surface tension support diminished, the hair, which is naturally denser than water, then sinks. Therefore, the Float Test is not measuring how much water the hair absorbs internally but rather how easily the hair’s surface interacts with and breaks the water’s surface tension. It’s a test of surface hydrophobicity/hydrophilicity, which is an indicator of surface damage, not internal porosity.
The Drop Test: Surface Spreading, Not Internal Absorption
The Drop Test involves placing a small drop of water onto a lock or section of hair and observing how it behaves. The conventional explanation posits that on "low porosity" hair, the water drop beads up and remains round, indicating sealed cuticles that repel water. On "high porosity" hair, the water drop is said to flatten out and absorb quickly, signifying open cuticles and rapid internal water uptake.

Again, the scientific explanation points to surface interactions rather than internal absorption. When a water drop is placed on an intact, undamaged hair surface (with its hydrophobic F-layer), the strong cohesive forces within the water drop itself (due to hydrogen bonding) are stronger than its adhesive forces to the hair surface. This causes the water to minimize its contact area with the hair, resulting in a spherical or beaded shape, maintaining high surface tension within the drop.
Conversely, on damaged hair, where the F-layer is compromised and the surface is more hydrophilic, the adhesive forces between the water molecules and the hair surface become stronger. The water molecules are more attracted to the hair surface than to each other, causing the drop to spread out and flatten. This spreading allows the water to cover a larger surface area of the hair, rather than being rapidly absorbed into the hair shaft. While damaged hair can absorb slightly more water (around 45% of its weight) compared to undamaged hair (30%), this difference is primarily due to increased access points for gaseous water (humidity) and not instantaneous liquid water absorption during a brief drop test. The observation of spreading is a phenomenon of wetting, where the liquid adheres to and spreads across a solid surface due to molecular attraction, again, indicating surface damage and hydrophilicity, not internal porosity.
Why "Porosity" Advice Sometimes Works (Coincidentally)
Despite the scientific inaccuracies of these DIY tests, it’s undeniable that many people find haircare advice based on their "porosity" diagnosis to be effective. This apparent paradox can be explained by the fact that the tests, while misinterpreting the underlying mechanism, do inadvertently assess a critical aspect of hair health: surface damage.
Hair that is frequently exposed to damaging processes like bleaching, coloring, perming, excessive heat styling, or harsh mechanical manipulation will inevitably suffer damage to its outer cuticle layer. This damage manifests as a compromised F-layer, lifted or chipped cuticle scales, and an overall more hydrophilic surface. Consequently, such hair will likely "test" as "high porosity" in the float and drop tests because its damaged surface readily interacts with water, reducing surface tension and causing spreading or sinking.

Conversely, hair that is relatively undamaged, perhaps virgin hair or hair that has been meticulously cared for, will retain a more intact F-layer and cuticle structure. This hair will likely "test" as "low porosity" because its hydrophobic surface resists immediate interaction with water, allowing it to float or bead water drops.
The product recommendations that follow these diagnoses often align with the actual needs of damaged versus healthy hair:
- "High porosity" advice: Often suggests richer, heavier conditioners, leave-ins, and oils. These products typically contain ingredients that can temporarily smooth damaged cuticles, provide a protective barrier, and help retain moisture that might otherwise escape from hair with compromised integrity. This aligns well with the needs of truly damaged hair.
- "Low porosity" advice: Often recommends lighter products, avoiding heavy butters and oils that might build up. This is suitable for healthier hair that doesn’t need intense conditioning and can become easily weighed down by excessive product.
Therefore, the "porosity" advice works not because the tests accurately measure internal absorption rates, but because they serve as a rough proxy for surface integrity and damage level. The correlation is coincidental but often beneficial.
Critical Implications for Haircare Professionals
While the coincidental effectiveness might suffice for individual consumers experimenting with products, the scientific misrepresentation of porosity has critical implications, particularly for professional hairdressers and stylists. Relying on these unscientific tests to guide complex chemical treatments can lead to suboptimal results or even further damage.
For example, chemical treatments such as coloring, perming, or straightening require specific processing times that depend on how quickly and effectively the chemicals can penetrate the hair shaft. The belief that "high porosity" hair (as diagnosed by DIY tests) means rapid internal absorption could lead a stylist to reduce processing times prematurely, resulting in incomplete chemical reactions and poor outcomes (e.g., uneven color, weak curls). Conversely, an "oily" or product-laden hair strand might give a "low porosity" reading, leading to unnecessarily prolonged processing times that could over-process and severely damage the hair.
The crucial point is that these tests are measuring surface hydrophobicity/hydrophilicity, which is influenced by the outermost layer of the hair and any product residue. This is distinct from the internal structure of the hair and its true capacity for chemical uptake, which is governed by factors like cuticle integrity, cortical density, and hair fiber diameter. A client with significant surface damage (diagnosed as "high porosity") might still have a dense internal structure that resists chemical penetration, requiring standard or even longer processing times.
For this reason, experts strongly advise against using the float or drop tests to determine processing times for chemical services. The most reliable method remains a strand test: directly applying the chemical product to a small, inconspicuous section of the client’s hair to observe its reaction and determine the appropriate timing. This empirical approach provides accurate, real-time information about how the specific hair strand will respond to the treatment, regardless of superficial characteristics.
Moreover, the prevalence of these myths can lead to consumer confusion and misdiagnosis. Someone with naturally fine hair might incorrectly assume it’s "low porosity" because it feels smooth, when in fact, its fine diameter makes it prone to damage and quicker saturation. Conversely, someone with coarse, virgin hair might believe it’s "high porosity" because it takes a long time to dry, a characteristic more related to hair density and thickness than cuticle openness.
Towards a More Scientific Approach to Hair Health

The debunking of popular hair porosity tests underscores the broader need for scientific literacy in the beauty industry. Instead of relying on misleading DIY methods, individuals and professionals should focus on a holistic understanding of hair health, taking into account various factors:
- Visual and Tactile Assessment: Observing the hair’s appearance (shine, frizz, split ends) and feeling its texture (roughness, smoothness, elasticity) can provide valuable insights into its condition.
- Hair History: Understanding past chemical treatments, heat styling habits, and environmental exposures is crucial for assessing damage.
- Professional Diagnosis: Consulting with a qualified trichologist or experienced hairdresser for a comprehensive assessment provides the most accurate evaluation of hair health.
- Ingredient Knowledge: Learning about the function of different haircare ingredients (humectants, emollients, proteins, film-formers) and how they interact with hair can lead to more informed product choices, irrespective of a "porosity" label.
In conclusion, while the term "hair porosity" may continue to be used in popular haircare discourse, it is imperative to understand that the common at-home tests are not scientifically valid indicators of internal hair porosity. They are, at best, rough diagnostics for surface damage and hydrophobicity. Moving forward, a more evidence-based approach, grounded in genuine hair science and professional expertise, will lead to more effective and truly personalized haircare solutions. The focus should shift from a simplistic "porosity" label to a nuanced understanding of hair’s unique structural characteristics, its history of treatments, and its ongoing environmental interactions.
References:
- Robbins CR. Chemical and Physical Behavior of Human Hair. 5th ed. Springer Berlin Heidelberg 2012.
- La Torre C,