A ubiquitous online beauty hack involving thermal paper receipts to assess the efficacy of heat protectants has been thoroughly debunked by scientific analysis, revealing that the test primarily measures chemical interactions with the receipt’s ink rather than genuine heat protection for hair. The viral method, which gained widespread traction on platforms like TikTok, suggested that heat protectants preventing thermal paper from darkening were superior, a premise now shown to be fundamentally flawed. Dr. Michelle Wong, a cosmetic chemist and founder of Lab Muffin Beauty Science, meticulously investigated the phenomenon, concluding that the observed "protection" is largely due to the solvent and surfactant properties of the beauty products, alongside the transient cooling effect of water.

The Rise of a Viral Beauty Test
The thermal paper receipt test emerged as a popular DIY experiment, particularly amplified through social media channels. The premise was deceptively simple and visually compelling: users would spray a heat protectant onto a section of a thermal paper receipt, then apply a hot flat iron. Thermal paper, commonly used for store receipts, turns black when exposed to heat, as its leuco dye coating reacts with a developer. The logic put forth by content creators, such as cosmetologist Lucy Seitz, was that a "working" heat protectant would insulate the paper, keeping it white, thereby implying similar protection for hair. This visual clarity and ease of replication made the test highly shareable and influential, leading many consumers to make purchasing decisions based on its misleading results. The immediate, dramatic visual contrast resonated with a public increasingly seeking quick, accessible validations for beauty claims, often overlooking the complex science involved.

However, scientific skepticism quickly arose regarding the validity of using thermal paper as a proxy for human hair. While models are often used in scientific research—from testing medications on mice to evaluating skincare on reconstructed human skin models—the critical question remains whether the model adequately mimics the real-world scenario. The initial appeal of the receipt test lay in the superficial similarity: both hair and thermal paper react to heat. Hair suffers structural damage from excessive heat, primarily protein denaturation and moisture loss, while receipt paper darkens due to a chemical reaction. The crucial missing link, as Dr. Wong’s investigation highlighted, was whether the conditions for these reactions were comparable and if the protective mechanisms translated across the two very different substrates.
Dr. Wong’s Comprehensive Investigation: Initial Observations

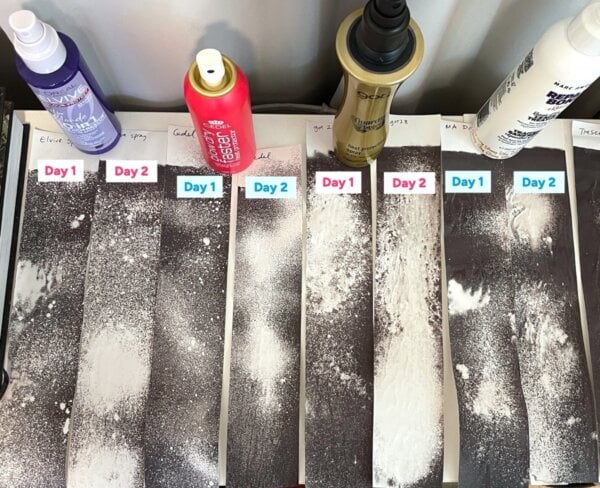
Dr. Michelle Wong embarked on a detailed scientific inquiry to scrutinize the viral test, documenting her process in both video and written formats. Her investigation began with an attempt to replicate the widely circulated tests, specifically those by Lucy Seitz, who had demonstrated a systematic approach by stating the straightener temperature (340°F or approximately 170°C) and using controls.
Initially, Dr. Wong observed several anomalies. When applying a heat protectant (Marc Anthony spray, one product also used by Seitz) to a receipt and immediately applying a straightener, it crackled and smoked. This suggested that residual moisture was a significant factor. Seitz herself had modified her tests to include a drying step after a commenter pointed out wet receipts, yet Dr. Wong noted that some receipts still appeared to be dripping. This led to an early hypothesis: the presence of water could be artificially "protecting" the receipts. Water is an excellent heat sink due to its high specific heat capacity and latent heat of vaporization. As it evaporates, it absorbs a substantial amount of thermal energy, effectively cooling the surface of the receipt and preventing the leuco dye from reacting.

To further explore this, Dr. Wong conducted her first series of experiments (Day 1) using 11 heat protectant products, including 7 pump sprays, 1 propellant spray, and 3 cream products. She applied these to receipts and allowed them to dry for approximately 15 minutes before applying heat. The results showed a clear distinction:
- Creams: These products generally resulted in the lightest receipts. Dr. Wong posited that creams, applied more thickly than sprays, would provide greater insulation and likely retain more moisture, drying slower than sprays.
- Sprays: The darkest receipts were observed with Goldwell and IGK sprays. Notably, these products had water listed lower on their ingredient lists, suggesting a lower water content and thus less cooling effect from evaporation.
- Drying Time: A stark difference was noted between receipts heated immediately after application and those allowed 15 minutes to dry, with the latter showing more darkening. This supported the hypothesis that water content played a crucial role in the immediate "protective" effect.
Other initial observations further complicated the picture: some receipts turned grey immediately after product application, even before heat was applied. This was an unexpected result, as heat protectants are not typically designed to interact with paper in this way. Moreover, some receipts appeared lighter after 24 hours of drying compared to their 15-minute counterparts, contradicting the expectation that a fully dry product would offer less evaporative cooling and thus lead to a darker receipt.

The Critical Discovery: Thermal Paper’s Temperature Threshold
A pivotal turning point in the investigation came when Dr. Wong delved into the chemistry of thermal paper. Her initial search for "thermal paper temperature" yielded misleading results, often citing activation temperatures similar to hair straighteners (150-185°C). However, a deeper dive, including a German Wikipedia article and scientific papers, revealed a critical piece of information: thermal paper begins to develop color at a much lower temperature, typically between 60 to 100°C (140-212°F), and reaches full density between 70 and 120°C (158-248°F).

This discrepancy was monumental. If thermal paper reacts at temperatures as low as 60°C, the test becomes irrelevant to hair damage, which generally occurs above 100°C, with significant damage from styling tools often seen at 170°C and above. To confirm this, Dr. Wong conducted "Receipt Temperature Tests" by dipping various receipts into water cooled to different temperatures from boiling. Her results consistently showed that most receipts turned black around 95°C, aligning the paper’s reaction threshold closer to the lower end of hair damage temperatures, but still leaving questions about the "protection" seen at higher straightener temperatures.
Unraveling the True Mechanism: Solvents and Surfactants

With the temperature discrepancy clarified, Dr. Wong revisited her observations that could not be explained by water evaporation alone. The phenomenon of receipts turning grey immediately upon product application, and some receipts appearing lighter after 24 hours of drying, pointed to a chemical interaction beyond simple heat transfer. She noticed that products causing immediate greying often had high alcohol content.
This led to a new, central hypothesis: Heat protectants that appear to "work" in this test are actually dissolving the ink layer on the receipt. Thermal paper is coated with a complex layer containing invisible leuco dyes, a developer, and often sensitizers, all embedded in a binder. When heated, the binder melts, allowing the dye and developer to mix and react, forming the visible black pigment. However, this ink layer is fragile. Solvents can disrupt this delicate structure.

Alcohol, a common ingredient in many spray-format beauty products, is a potent solvent. It can dissolve the oily components of the thermal ink layer, prematurely reacting the dye or, more likely, disrupting the arrangement of the dye and developer so they cannot properly react to form a dark image when heat is applied. This also explains why diluted alcohol might work better than pure alcohol: water and humectants (like glycerin) slow down the evaporation of alcohol, allowing it more time to interact with and disrupt the ink layer.
To confirm this "dissolving" hypothesis, Dr. Wong performed "Confirmatory Receipt Tests" using various common substances:

- Alcohol, Water, Glycerin: Drops of methylated spirits, alcohol mixed with water, and alcohol with water and glycerin all kept receipts light after 30 minutes of drying and heating. Diluted alcohol produced a whiter result than pure alcohol, supporting the idea that slower evaporation due to water and glycerin allowed for more ink disruption. Water and glycerin alone, being less effective solvents for the oily ink components, did not significantly disturb the ink.
- Other Alcohol-Containing Products: Perfume, which is high in alcohol, immediately turned receipts grey and kept them white upon heating. Dry shampoo, also alcohol-rich, showed similar immediate greying but less "protection" upon heating, possibly due to faster alcohol evaporation with fewer humectants.
- Surfactants: Many heat protectants that "worked" but lacked high alcohol content contained surfactants (emulsifiers). Surfactants are molecules that reduce surface tension and are excellent at dissolving oily substances. Tests with micellar water and water with detergent demonstrated that these also kept receipts white after 30 minutes. This extends the hypothesis: not just alcohol, but any strong solvent or surfactant present in a heat protectant can interfere with the thermal paper’s ink. This also explained why creams, rich in emulsifiers (surfactants), often yielded very white results.
- Volatile Silicones: Cyclopentasiloxane, a volatile silicone commonly found in hair products, had minimal impact on receipts, likely because it is too oily to dissolve the ink and evaporates too quickly. This explained why some silicone-based products "failed" the receipt test, despite being effective heat protectants for hair.
- Propellants: Compressed air (primarily HFC-152a propellant) had no effect, indicating propellants themselves were not the cause of ink disruption.
Further validation came from Dr. Wong’s "Erasing Receipt Ink" experiment. She found that applying alcohol, perfume, sunscreen, micellar water, and moisturizer to already darkened thermal paper could largely erase the black print. This demonstrated that these substances could actively break up the formed ink pigment, directly supporting the idea that they interfere with the thermal paper’s chemical reaction.
The True Science of Heat Protection for Hair

The findings definitively show that the viral receipt test is an unreliable indicator of a product’s ability to protect hair from heat damage. Heat protectants for hair do not work by "blocking" heat in the way an insulator would. Instead, their primary mechanisms include:
- Even Heat Distribution: Ingredients like silicones (e.g., dimethicone, amodimethicone) and polymers (e.g., PVP/VA copolymers) form a thin, smooth film on the hair shaft. This film helps to distribute heat more evenly across the hair strand, preventing localized "hot spots" that can cause severe damage.
- Reducing Friction: The smooth film also reduces friction between the hair and hot styling tools, minimizing mechanical damage like snagging, breakage, and cuticle lifting.
- Moisture Retention: Some ingredients, like hydrolyzed proteins (e.g., hydrolyzed wheat protein, hydrolyzed soy protein) and humectants, can help retain moisture within the hair, making it more resilient to the drying effects of heat.
- Heat Absorption/Dissipation: Certain polymers can absorb some of the heat energy, acting as sacrificial layers that break down before the hair’s keratin structure.
Crucially, applying water-based products to hair immediately before heat styling is detrimental. As Dr. Wong noted, residual water in the hair can explosively evaporate when heated by a hot tool, leading to internal structural damage known as "bubble hair," where steam pockets form within the hair shaft, causing severe weakening and breakage. This directly contradicts the false "protection" observed in wet receipt tests.
Legitimate beauty brands conduct rigorous instrumental and clinical testing on actual hair samples to validate heat protection claims. For example, Amika, a brand whose product "failed" the receipt test, publicly shared footage of their testing process. This often involves specialized equipment that simulates styling, such as machines that repeatedly comb treated hair samples at high temperatures, followed by microscopic analysis to assess cuticle integrity, breakage, and tensile strength. Consumers should look for product claims that reference specific temperatures, instrumental tests, or comparative studies (e.g., "vs. non-conditioning shampoo alone") as indicators of scientifically validated protection.
Implications for Consumers and Beauty Science in the Digital Age

The debunking of the viral heat protectant receipt test serves as a crucial case study in the broader challenge of discerning scientific accuracy amidst a deluge of online beauty content. The ease with which visually convincing but scientifically unsound "hacks" can spread highlights the need for critical media literacy among consumers.
The implications are far-reaching:

- Consumer Misinformation: The test has likely led countless individuals to believe ineffective products were protecting their hair, potentially causing long-term damage from heat styling. Conversely, genuinely effective products might have been dismissed because they "failed" the flawed receipt test.
- Brand Reputation: Brands whose products "failed" the test due to their formulation (e.g., containing volatile silicones that don’t dissolve thermal ink, or simply having less water) faced unfair criticism, while those with high solvent content inadvertently appeared superior. This underscores the difficulty for brands to counter viral misinformation with complex scientific explanations.
- The Role of Cosmetic Chemists: The investigation by Dr. Wong exemplifies the vital role of trained scientists in clarifying complex beauty chemistry and challenging unsubstantiated claims. It emphasizes that real beauty science often involves intricate processes and specialized equipment, far removed from simple kitchen experiments.
- Education and Advocacy: This incident reinforces the ongoing need for consumer education on how beauty products truly function and the importance of seeking information from credible, scientifically literate sources. It encourages a shift from relying on anecdotal visual evidence to understanding the underlying scientific principles.
In conclusion, the viral heat protectant receipt test, while visually engaging, is a misleading indicator of hair protection. Its results are primarily dictated by the chemical interaction of product ingredients (like solvents and surfactants) with the thermal paper’s delicate ink layer, and the transient cooling effect of water, none of which accurately reflect how heat protectants safeguard human hair. For genuine protection, consumers should trust products backed by rigorous scientific testing on actual hair, understanding that effective heat protectants work by distributing heat, reducing friction, and maintaining hair integrity, rather than simply "blocking" heat.
