For years, the concept of "hair porosity" has been a cornerstone of popular haircare advice, guiding countless individuals toward products and routines supposedly tailored to their hair’s unique absorption capabilities. However, a recent in-depth analysis by Lab Muffin Beauty Science, published on January 28, 2026, unequivocally debunks the scientific basis of widely circulated at-home porosity tests, revealing that these methods often misinterpret fundamental principles of hair science. Authored by Michelle Wong, this exposé asserts that popular tests like the "Float Test" and "Drop Test" do not measure hair’s internal porosity or water absorption rates, but rather its surface interaction with water, a phenomenon primarily governed by surface tension.
The pervasive notion that hair porosity dictates how well hair absorbs moisture has led to a complex taxonomy of "low," "medium," and "high" porosity hair, each with its own recommended product categories and care strategies. While many individuals report finding suitable products through this framework, the underlying scientific explanations for these tests are largely coincidental and, in many cases, fundamentally flawed. The article, adapted from Wong’s video on hair hydration and building on previous discussions about hair and water and the myth of hygral fatigue, aims to dismantle these misconceptions and provide a more accurate, science-backed understanding of hair’s interaction with water.
The Myth of Waterproof Hair and Flawed Porosity Tests
A central misconception fueling the porosity myth is the belief that undamaged hair is inherently waterproof. This idea suggests that the hair’s outermost layer, the cuticle, forms an impenetrable barrier to water, and that conditioners serve to mimic this sealing function, particularly for damaged hair. Lab Muffin Beauty Science challenges this notion directly, presenting compelling evidence that even pristine, healthy hair readily absorbs significant amounts of water.

According to research cited from Robbins CR’s "Chemical and Physical Behavior of Human Hair" (5th ed., 2012), undamaged human hair can absorb nearly one-third (approximately 30%) of its own weight in water within minutes. This absorption is dynamic, with the water content of hair rapidly adjusting to ambient humidity levels. The following table illustrates this responsiveness:
| Relative humidity (%) | Weight of water absorbed (%) |
|---|---|
| 0 | 0 |
| 8 | 3.9 |
| 40 | 10.2 |
| 63 | 14.8 |
| 86 | 22.6 |
| 100 | 31.2 |
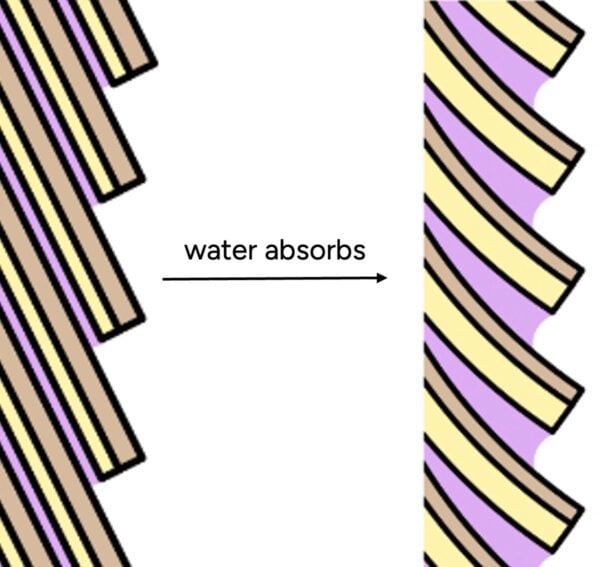
This data clearly demonstrates that hair, far from being waterproof, is highly hygroscopic, meaning it readily absorbs moisture from the air. The reason for this lies in the intricate structure of the hair cuticle. Unlike a seamless, sealed surface, the cuticle is composed of overlapping scales, often compared to shingles on a roof or the scales of a pinecone. The hair’s natural conditioning "F-layer," a thin, fatty acid layer that makes hair hydrophobic, is present only on the top of each cuticle scale. This leaves numerous microscopic gaps and edges where water molecules can penetrate and enter the hair shaft.
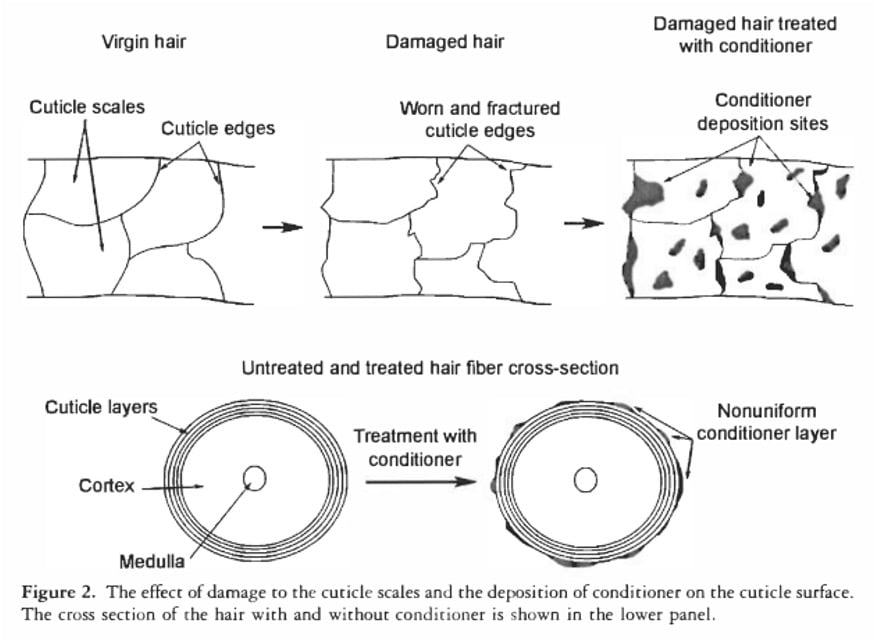
Furthermore, the notion that conditioners "seal" the hair cuticle to repel water is also inaccurate. Scientific studies, such as that by La Torre C and Bhushan B (J Cosmet Sci. 2006;57(1):37-56), demonstrate that conditioners deposit onto the hair surface not as a continuous, impermeable film, but rather as microscopic "blobs" or patches. While these deposits are effective at smoothing the hair’s surface, reducing friction, and imparting a soft feel, they are not robust enough to create a barrier against tiny water molecules. The individual water molecules are significantly smaller than these conditioner deposits, allowing them to easily bypass the "blobs" and access the hair shaft.
These scientific realities directly contradict the premises of popular at-home porosity tests:
The Float Test: This test instructs individuals to place a single strand of hair in a glass of water. The interpretation posits that "high porosity" (damaged) hair will sink rapidly because its compromised cuticle allows water to quickly absorb, increasing its density. Conversely, "low porosity" (undamaged) hair is believed to float due being waterproof and resisting absorption.
The Drop Test: In this test, a drop of water is placed on a lock of hair. The prevailing belief is that on undamaged, "low porosity" hair, the water will bead up and remain as a round droplet, indicating poor absorption. On damaged, "high porosity" hair, the water drop is expected to flatten out and quickly disappear, signifying rapid absorption into the hair.
Both tests are presented as direct indicators of how quickly hair absorbs water internally. However, as Lab Muffin’s analysis highlights, their interpretations are fundamentally flawed because they overlook the critical role of surface phenomena.
Surface Tension: The True Explainer
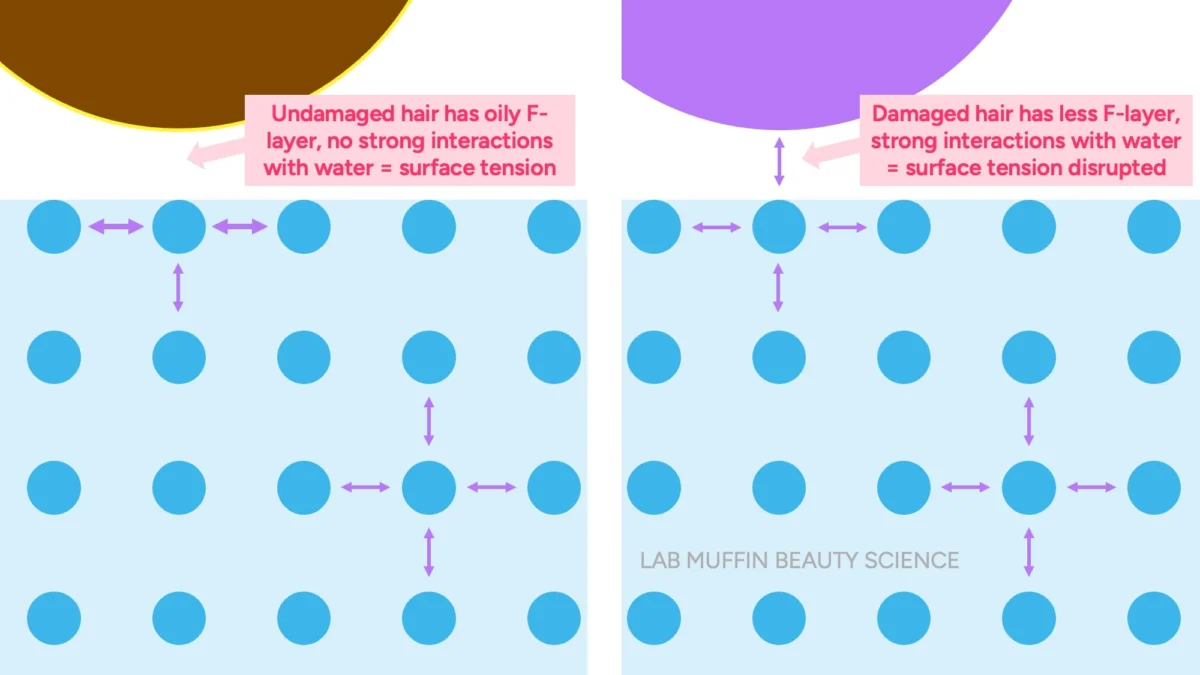
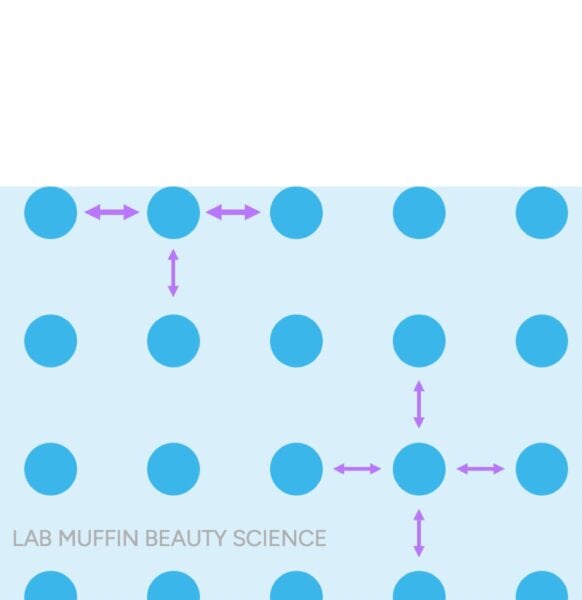
The key to understanding why these tests produce their observed results, despite their incorrect interpretations, lies in the principle of surface tension. Surface tension is a physical property of liquids that arises from the cohesive forces between molecules at the liquid’s surface. In water, individual water molecules are strongly attracted to each other through hydrogen bonds. Molecules within the bulk of the liquid are surrounded by other water molecules on all sides, forming a balanced network of attractions. However, water molecules at the surface lack neighbors above them, leading to stronger inward pulls from adjacent and underlying molecules. This creates a net inward force, effectively forming a "skin" or elastic film on the liquid’s surface.
This "skin" is remarkably strong, capable of supporting objects denser than water, such as insects, small paperclips, or even a strand of hair, preventing them from sinking. The classic demonstration involves carefully placing a steel paperclip (which is roughly eight times denser than water) on the water’s surface; it floats due to surface tension. If this surface tension is disrupted—by poking the paperclip, adding a drop of detergent (a surfactant that reduces surface tension), or by the object itself interacting strongly with water—the paperclip immediately sinks.
Explaining the Float Test through Surface Tension:
Applying the principle of surface tension, the Float Test takes on a new scientific meaning. Undamaged hair possesses an intact F-layer on its cuticle surface. This F-layer is naturally hydrophobic, meaning it repels water. When an undamaged hair strand is placed on water, its hydrophobic surface resists forming strong hydrogen bonds with the water molecules at the surface. Consequently, the water’s surface tension remains largely intact beneath the hair strand, allowing the hair to "float" on this invisible skin, much like a paperclip.
Conversely, damaged hair has a compromised or absent F-layer, exposing a more hydrophilic (water-loving) surface. When damaged hair encounters water, its surface readily forms hydrogen bonds with the water molecules, effectively "holding hands" with them. This interaction disrupts the cohesive network of water molecules at the surface, weakening the surface tension. Once the surface tension is sufficiently reduced, the hair strand, which is intrinsically denser than water, loses its support and sinks. Therefore, the Float Test is not measuring the rate of water absorption into the hair shaft, but rather the integrity of the hair’s hydrophobic F-layer and its impact on the water’s surface tension.
Explaining the Drop Test through Surface Tension:
The Drop Test is similarly explained by surface tension and the surface properties of hair, not by internal absorption. When a drop of water is placed on undamaged hair, the hydrophobic F-layer minimizes contact and adhesion between the water molecules and the hair surface. The strong cohesive forces within the water droplet itself (its own surface tension) cause it to bead up into a spherical shape, resisting flattening and spreading.

On damaged hair, the exposed hydrophilic surface readily interacts with the water drop. This strong attraction between the water molecules and the hair surface overcomes the water’s internal cohesive forces, causing the droplet to flatten out and spread across the hair surface. The water is not necessarily "absorbing" into the hair shaft faster in its liquid form, but rather spreading along the damaged, water-attracting surface of the hair and between individual strands in the lock. It is crucial to remember that even damaged hair absorbs only slightly more water (around 45% of its weight) than undamaged hair (30%), making rapid liquid absorption an unlikely primary explanation for the immediate flattening observed in the Drop Test.
The Role of Gaseous Water Absorption:
So, if liquid water doesn’t rapidly soak into undamaged hair due to "porosity" in these tests, how does undamaged hair absorb 30% of its weight in water, as the Robbins data shows? The answer lies in the state of the water: it’s gaseous water (humidity), not liquid water. In humid air, individual water molecules are not bound together by strong surface tension. These solitary, tiny gas molecules are small enough to easily wiggle through the microscopic gaps between the cuticle scales and penetrate the hair shaft, without needing to overcome the cohesive forces of liquid water. This distinction is vital for a scientific understanding of hair hydration.
Why "Porosity" Advice Often Works (Despite Flawed Tests)
While the scientific premise of these tests is incorrect, it’s important to acknowledge why the resulting haircare advice often appears effective. Lab Muffin Beauty Science clarifies that these tests, while mislabeled, do serve as proxies for surface damage. A "high porosity" result, according to these tests, indicates significant damage to the hair’s surface, particularly the F-layer. Conversely, a "low porosity" result suggests a more intact and less damaged surface.

Haircare strategies based on surface damage are, for the most part, entirely sensible. Hair with a compromised cuticle (diagnosed as "high porosity" by these tests) often benefits from more intensive moisturizing treatments, protein-rich products to help temporarily patch up damaged areas, and protective styling to prevent further damage. This is because a damaged cuticle can lead to increased frizz, dryness, and vulnerability. Hair with an intact cuticle (diagnosed as "low porosity") may require lighter products to avoid buildup and might be more prone to product accumulation due to its smoother surface.
Therefore, the tests, by indirectly assessing surface damage, coincidentally guide users toward products that address the actual condition of their hair’s outer layer. The "porosity" label simply provides an incorrect scientific justification for what is essentially a response to varying degrees of surface integrity.
Implications and Professional Responsibility
The debunking of hair porosity tests carries significant implications for both consumers and haircare professionals. For consumers, understanding the true science empowers them to make more informed decisions, reducing reliance on potentially misleading diagnostic tools. It shifts the focus from an abstract concept of "porosity" to the more tangible reality of "surface integrity" and "damage," which can be addressed with appropriate care.
For hairdressers and salon professionals, the stakes are even higher. The article issues a strong warning: do not rely on these porosity tests to determine the processing time for chemical treatments. Chemical services such as coloring, perming, or straightening depend critically on the hair’s internal structure and its ability to absorb and react with chemical agents. While surface damage might affect how quickly chemicals initially penetrate the outer layers, it doesn’t accurately predict the overall rate or extent of chemical processing throughout the hair shaft. Misjudging processing times based on these flawed tests can lead to disastrous results, including over-processing, severe damage, or ineffective treatments. The scientifically sound method remains a direct strand test with the actual product to gauge individual hair’s response.
A Call for Scientific Literacy in Beauty
The persistence of myths like the "porosity tests" highlights a broader challenge within the beauty industry: the gap between popular lore and scientific fact. In an age where information is abundant but often unchecked, science communicators like Michelle Wong of Lab Muffin Beauty Science play a crucial role in providing accurate, evidence-based insights. By meticulously dissecting common misconceptions and offering clear, accessible explanations rooted in chemistry and physics, they empower consumers to navigate the complex world of haircare with greater confidence and understanding.
The scientific community continues to refine its understanding of hair structure and its interactions with various compounds. While the term "porosity" may hold a more complex, nuanced meaning in advanced trichology, its colloquial application via simple at-home tests has proven to be a misdirection. The message from Lab Muffin Beauty Science is clear: while hair health is undoubtedly a vital concern, the methods used to assess it must align with established scientific principles. Moving forward, both consumers and professionals are encouraged to prioritize evidence-based knowledge over widespread, yet scientifically unfounded, practices.
Affiliate Disclosure: I receive a small commission for purchases made via affiliate links.
How to cite: Wong M. Hair porosity tests are a lie. Lab Muffin Beauty Science. January 28, 2026. Accessed March 30, 2026. https://labmuffin.com/hair-porosity-tests-are-a-lie/
References:
- Robbins CR. Chemical and Physical Behavior of Human Hair. 5th ed. Springer Berlin Heidelberg 2012.
- La Torre C, Bhushan B. Nanotribological effects of silicone type, silicone deposition level, and surfactant type on human hair using atomic force microscopy. J Cosmet Sci. 2006;57(1):37-56.