The concept of "hair porosity" has become a cornerstone of modern haircare advice, guiding countless consumers to select products based on whether their hair is deemed "low," "medium," or "high" porosity. This complex term, often simplified to describe how easily hair absorbs and retains moisture, has led to a proliferation of do-it-yourself tests designed to help individuals determine their hair’s specific needs. However, a growing body of scientific research, spearheaded by cosmetic chemists and hair scientists, indicates that these widely popular diagnostic methods are fundamentally flawed, relying on misconceptions about hair’s interaction with water. While some porosity-based advice may coincidentally lead to beneficial product choices, the scientific premise underlying these common tests, such as the float test and the drop test, is largely inaccurate, attributing phenomena to internal absorption when external surface properties are the true drivers.
The Rise of "Hair Porosity" in Beauty Culture
The term "hair porosity" gained significant traction within the beauty community, particularly among those with textured and curly hair, as individuals sought to better understand and care for their unique hair types. The prevailing understanding posits that hair porosity is determined by the condition of the hair’s outermost layer, the cuticle. A tightly bound, smooth cuticle is said to indicate "low porosity," making it difficult for water and products to penetrate but easy to retain moisture. Conversely, a raised or damaged cuticle is associated with "high porosity," allowing for rapid water absorption but also quick moisture loss. "Medium porosity" falls somewhere in between, representing balanced moisture dynamics.

This framework spurred the development of accessible, at-home tests. The most ubiquitous is the Float Test, where a strand of hair is placed in a glass of water, with the theory being that "high porosity" hair, being more absorbent, will quickly sink, while "low porosity" hair will float due to its water-repelling cuticle. Similarly, the Drop Test involves placing a water droplet on a section of hair; a quickly absorbed or flattened droplet signifies "high porosity," while a beaded droplet that remains on the surface points to "low porosity." These tests, appealing in their simplicity and directness, quickly became go-to diagnostics for millions seeking personalized haircare routines, influencing product formulations and marketing strategies across the industry.
Understanding Hair’s Interaction with Water: Beyond "Waterproof"
A core misconception underpinning the popular porosity tests is the idea that healthy, undamaged hair is inherently waterproof or acts as an impermeable barrier to water. Scientific investigation, however, paints a different picture. Hair, irrespective of its condition, is naturally hygroscopic, meaning it readily absorbs moisture from its environment. Research demonstrates that even undamaged hair can absorb a substantial amount of water—up to 30% to 31% of its own weight—within minutes. This absorption is not a slow, gradual process but a rapid exchange that occurs as hair seeks equilibrium with the surrounding humidity.
Data from seminal works in hair science, such as Robbins’ Chemical and Physical Behavior of Human Hair, illustrate this dynamic relationship:
| Relative Humidity (%) | Weight of Water Absorbed (%) |
|---|---|
| 0 | 0 |
| 8 | 3.9 |
| 40 | 10.2 |
| 63 | 14.8 |
| 86 | 22.6 |
| 100 | 31.2 |
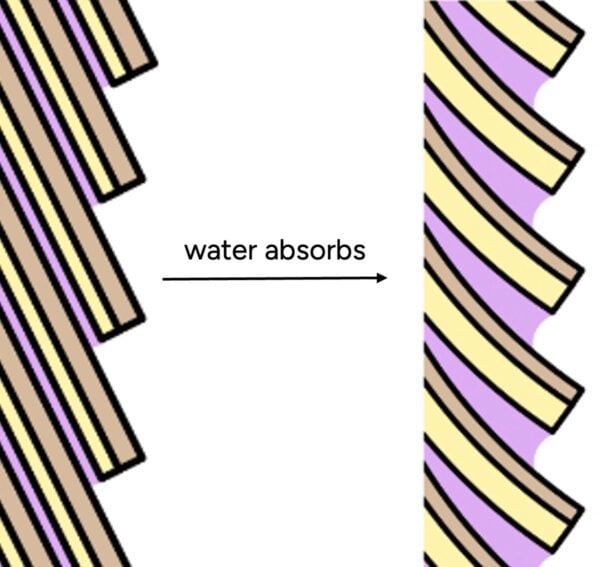
This table highlights that hair’s water content fluctuates significantly and quickly based on ambient humidity, demonstrating its intrinsic ability to absorb moisture. This absorption occurs because hair’s structure, often compared to a pinecone, is not perfectly sealed. The cuticle, composed of overlapping scales, presents numerous microscopic gaps where water molecules can penetrate. Furthermore, hair possesses a natural conditioning layer known as the F-layer (18-methyl eicosanoic acid), which lies on the surface of each cuticle scale. While the F-layer imparts a hydrophobic (water-repelling) quality, it is not a continuous, unbroken seal, leaving ample opportunities for water ingress.
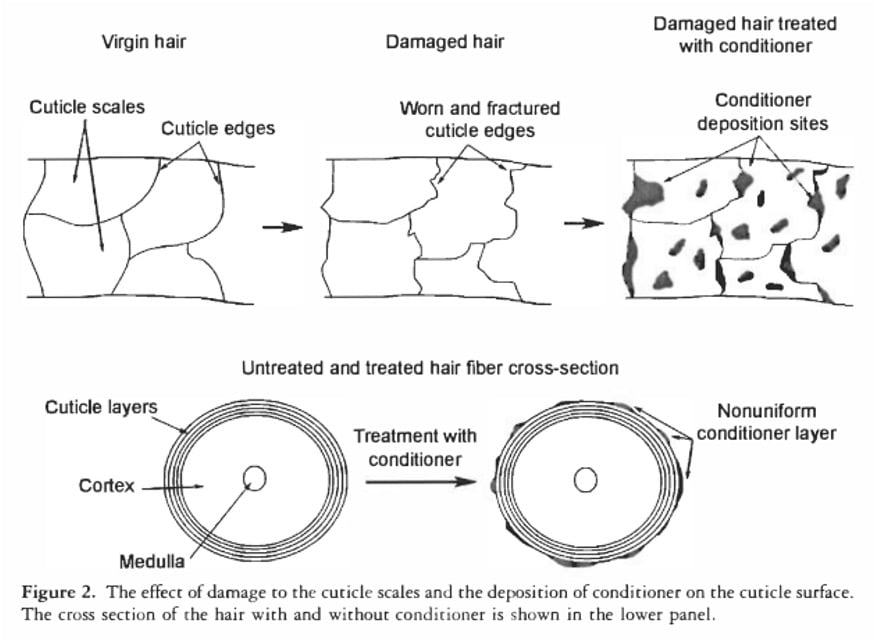
Even synthetic conditioners, which are designed to smooth the hair’s surface and reduce friction, do not render hair waterproof. Microscopic analyses, such as those conducted by La Torre and Bhushan, reveal that conditioner molecules deposit on the hair surface in "blobs" rather than forming a uniform, impenetrable film. While these molecular depositions are effective in making hair feel smooth and soft to the touch, they are insufficient to block the passage of individual water molecules, which are vastly smaller. Therefore, the notion that conditioners or an intact cuticle "seal out" water is scientifically unfounded; hair is designed to interact with and absorb water.
Debunking the DIY Diagnostics: The Role of Surface Tension
If undamaged hair is not waterproof and readily absorbs moisture, what then explains the observations made during the float and drop tests? The scientific explanation lies not in the hair’s internal absorption rate of liquid water but in the phenomenon of surface tension.
Surface tension is a property of liquid surfaces that causes them to behave like an elastic "skin." In water, this effect is particularly pronounced due to the strong cohesive forces (hydrogen bonds) between water molecules. Molecules within the bulk of the water are equally attracted in all directions by their neighbors, but those at the surface experience a net inward pull, creating tension. This "skin" is strong enough to support objects denser than water, such as insects, paperclips, or even a strand of hair, allowing them to "float" on the surface without sinking. A classic demonstration involves carefully placing a steel paperclip on water; despite steel being approximately eight times denser than water, it can float until the surface tension is disrupted, for example, by adding a drop of detergent.
Applying this principle to the hair porosity tests reveals their true mechanism:
-
Explaining the Float Test: When a strand of hair is placed on water, its initial behavior is dictated by the interaction between the hair’s surface and the water’s surface tension.
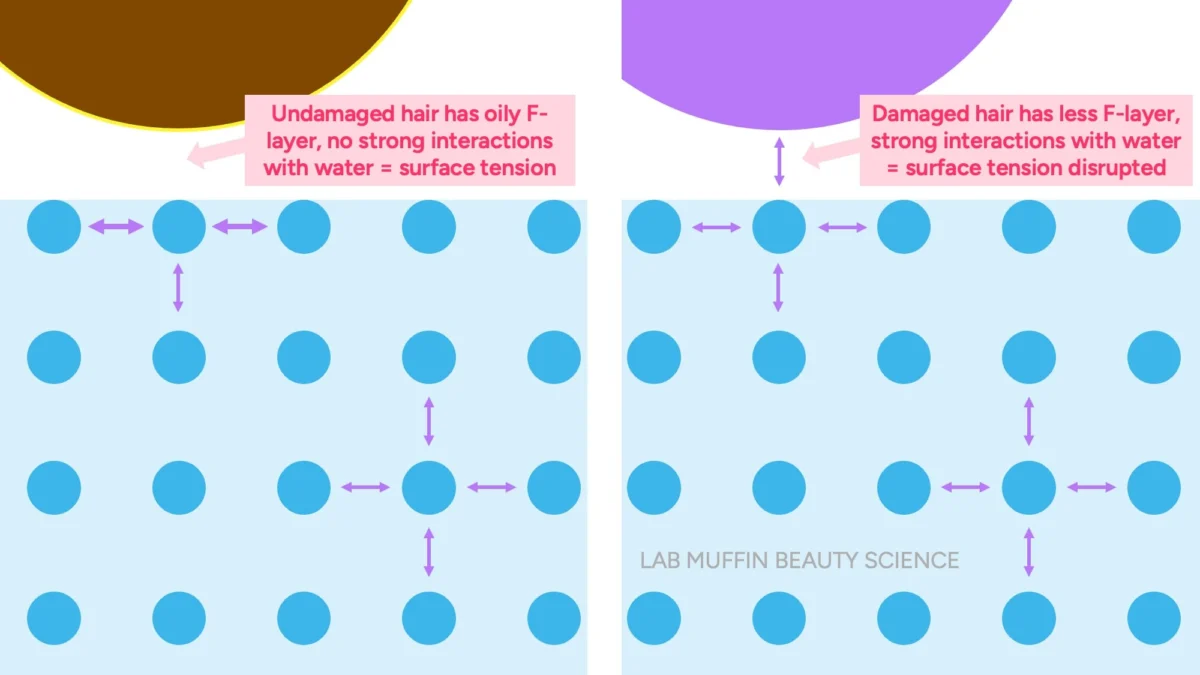
- Undamaged Hair: Possesses an intact F-layer, which is naturally hydrophobic. This oily layer helps to maintain the water’s surface tension, preventing the water from "wetting" the hair thoroughly. Consequently, the hair strand can be supported by the water’s surface tension and appears to "float."
- Damaged Hair: Has a compromised or removed F-layer due to chemical treatments, heat styling, or mechanical stress. This exposes a more hydrophilic (water-loving) keratin surface. When this damaged surface meets water, it forms hydrogen bonds with the water molecules, effectively disrupting the water’s surface tension at the contact point. With the supporting "skin" weakened, the hair strand, which is denser than water, will sink.
The key takeaway is that the sinking of damaged hair is primarily a consequence of its surface disrupting water’s cohesive forces, not because it has rapidly absorbed a massive amount of liquid water internally to increase its density.
-
Explaining the Drop Test: Similar to the float test, the appearance of water droplets on hair is governed by surface tension and the hair’s surface chemistry, specifically its wettability or contact angle.

- Undamaged Hair: With its intact, hydrophobic F-layer, resists "wetting." A water droplet placed on its surface will maintain a high contact angle, appearing as a round bead, because the water molecules prefer to bond with each other rather than spread across the hair.
- Damaged Hair: Lacking a continuous F-layer and presenting a more hydrophilic surface, readily allows water to spread. The water droplet will exhibit a low contact angle, flattening out and appearing to absorb quickly, as the water molecules are more attracted to the hair’s surface.
Here again, the "absorption" observed in the drop test is primarily a phenomenon of surface spreading, not a rapid internal soaking of liquid water into large pores. The actual increase in internal water content between damaged and undamaged hair is significant but not as dramatically different as the tests imply, with damaged hair absorbing around 45% of its weight compared to 30-31% for undamaged hair. This subtle difference is largely due to the exposure of more internal sites for water binding, rather than large "holes" that quickly "suck in" liquid water.
It is crucial to differentiate between the absorption of liquid water and water vapor. While liquid water’s interaction with hair is heavily influenced by surface tension, water molecules in a gaseous state (humidity) are small enough to readily diffuse between cuticle scales and into the hair’s cortex, without encountering the same surface tension barriers. This explains why hair’s internal moisture content changes rapidly with humidity, regardless of surface damage.
Why "Porosity" Advice Often Seems to Work (and Where it Fails)
Despite the scientific debunking of the DIY porosity tests, many individuals report positive results from following "porosity-based" haircare advice. This apparent success is largely coincidental, as the tests inadvertently serve as indicators of hair surface damage rather than true internal porosity for liquid water.
- "High Porosity" as an Indicator of Damage: Hair that "sinks" or "spreads water" in the tests is typically hair with a damaged F-layer and lifted cuticle scales. This type of hair does benefit from products often recommended for "high porosity": heavier conditioners, protein treatments, and occlusive ingredients (like certain oils or butters). These ingredients work not by filling "holes" but by depositing on the surface to smooth the cuticle, reduce friction, and provide a protective barrier, thereby mitigating the effects of damage and helping to retain moisture that has already been absorbed.
- "Low Porosity" as an Indicator of Health or Product Buildup: Hair that "floats" or "beads water" often has a relatively intact F-layer, indicating less surface damage. This hair typically benefits from "low porosity" recommendations: lighter products, humectants, and occasionally heat to aid penetration. Sometimes, however, "low porosity" can also be an artifact of significant product buildup (e.g., heavy oils, silicones) that create an artificially hydrophobic surface, leading to a misleading test result.
The critical distinction is that the tests diagnose a symptom (surface wettability, indicative of damage or buildup) and not the underlying cause (an inherent, unchangeable "porosity" for liquid water). The haircare solutions recommended for these "porosity" types often align with what damaged or healthy hair actually needs, creating a perceived validation of the flawed diagnostic.

Implications for Professional Hairdressers and the Industry
For professional hairdressers, relying on these DIY porosity tests to inform critical chemical treatments (such as coloring, perms, or relaxers) can lead to suboptimal or even damaging results. Chemical penetration into the hair’s cortex is a complex process influenced by the hair’s internal structure, disulfide bonds, and the specific chemistry of the treatment product, not solely by the surface’s wettability or perceived "porosity." A strand of hair that tests as "high porosity" due to surface damage might not necessarily absorb chemical solutions more quickly or deeply than a strand that tests as "low porosity." Misjudging processing times based on these tests could result in under-processing, over-processing, or uneven results.
Leading hair scientists and industry experts advocate for direct strand tests using the actual chemical product as the only reliable method for determining processing times. This approach accounts for the unique chemical and structural properties of an individual’s hair and its specific interaction with the professional treatment.
The Broader Implications: Science Literacy in Beauty
The widespread adoption and subsequent debunking of hair porosity tests underscore a broader challenge in the beauty industry: the prevalence of pseudo-science and "science-washing." Consumers, eager for personalized solutions, are often susceptible to simplified explanations and easily performed tests that appear scientific. This highlights the urgent need for greater scientific literacy among consumers and enhanced transparency and accuracy from haircare brands and educators.
Moving forward, the focus in haircare science is shifting towards understanding hair’s actual structural integrity, its response to environmental factors like humidity, and the specific chemical interactions with product ingredients. Rather than relying on misleading "porosity" labels, consumers and professionals are encouraged to consider the holistic condition of the hair—its level of damage, its natural texture, its density, and its response to various products and environmental conditions—to make truly informed haircare decisions. Embracing evidence-based science over simplified myths empowers individuals to better care for their hair, fostering healthier practices and more effective product choices in the long run.