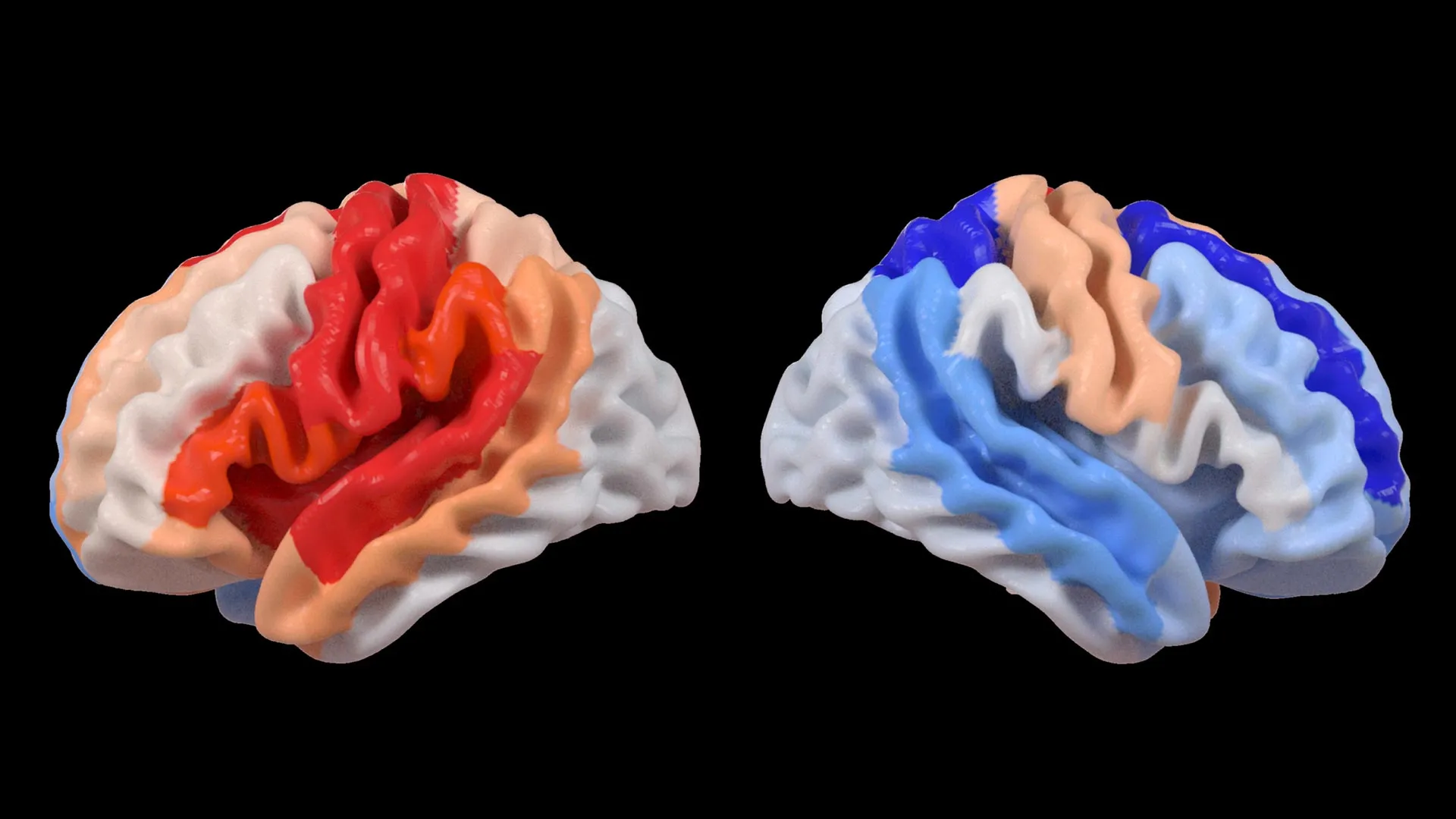
A groundbreaking new study published in the prestigious journal The Lancet Digital Health is shedding light on the brain’s remarkable ability to adapt following a stroke, revealing a surprising phenomenon: areas of the brain not directly affected by the injury may exhibit signs of being "younger" in individuals with severe physical impairments. Researchers at the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have utilized advanced artificial intelligence techniques to analyze a vast international dataset, uncovering patterns of brain reorganization that were previously undetectable. This discovery offers a novel perspective on neuroplasticity and holds significant promise for refining stroke recovery strategies.
Unveiling the Brain’s Counterintuitive Response to Injury
The study, a product of the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, represents a monumental collaborative effort. Scientists meticulously analyzed brain scans from over 500 stroke survivors, with data meticulously collected from 34 research centers spanning eight different countries. This extensive, multi-site approach is crucial for ensuring the generalizability of findings and overcoming the limitations of smaller, single-institution studies. By employing deep learning models, specifically a graph convolutional network, trained on tens of thousands of magnetic resonance imaging (MRI) scans, the research team was able to estimate the "brain age" of individual regions within each hemisphere of the brain. This innovative method allowed them to assess how stroke impacts not only the damaged areas but also the functional integrity and potential for recovery in seemingly unaffected regions.
The core finding is as intriguing as it is significant. While larger strokes were found to accelerate the aging process in the damaged hemisphere, the opposite hemisphere of the brain in these same individuals surprisingly appeared "younger." This phenomenon, as explained by Hosung Kim, PhD, associate professor of research neurology at the Keck School of Medicine of USC and co-senior author of the study, suggests a dynamic process of brain adaptation. "This pattern suggests the brain may be reorganizing itself, essentially rejuvenating undamaged networks to compensate for lost function," Dr. Kim stated. This counterintuitive observation challenges traditional understandings of stroke’s impact, highlighting the brain’s inherent resilience and its capacity for compensatory mechanisms.
AI as a Window into Brain Rewiring
The application of artificial intelligence, particularly a graph convolutional network, was instrumental in deciphering these complex brain patterns. This sophisticated AI system was trained to estimate the biological age of 18 distinct brain regions based on detailed MRI data. By comparing this AI-predicted age with each participant’s actual chronological age, researchers calculated a "brain-predicted age difference" (brain-PAD). This metric serves as a valuable indicator of overall brain health, with a negative brain-PAD suggesting a younger-than-expected biological age for a given region.
The study then correlated these brain age measurements with objective assessments of motor function. A striking correlation emerged: stroke survivors who experienced severe movement impairments, even after more than six months of intensive rehabilitation, exhibited a significantly younger-than-expected brain age in regions located on the side of the brain opposite to the stroke’s origin. This effect was particularly pronounced within the frontoparietal network, a critical brain circuit involved in a multitude of higher-order functions including movement planning, attention, and complex coordination.
Dr. Kim further elaborated on the implications of these findings: "These findings suggest that when stroke damage leads to greater movement loss, undamaged regions on the opposite side of the brain may adapt to help compensate. We saw this in the contralesional frontoparietal network, which showed a more ‘youthful’ pattern and is known to support motor planning, attention, and coordination. Rather than indicating full recovery of movement, this pattern may reflect the brain’s attempt to adjust when the damaged motor system can no longer function normally. This gives us a new way to see neuroplasticity that traditional imaging could not capture." This insight suggests that the "youthful" appearance of these contralateral regions may not signify a return to pre-stroke function but rather an adaptive response, a form of neural rewiring to mitigate the functional deficits caused by the initial injury.
The Power of Large-Scale Data in Revealing Hidden Patterns
The success of this study is inextricably linked to the ENIGMA initiative, a global collaborative network that aggregates data from over 50 countries. ENIGMA’s mission is to foster a deeper understanding of the human brain across a wide spectrum of neurological conditions. By establishing standardized protocols for MRI data acquisition and clinical information collection, the ENIGMA Stroke Recovery Working Group was able to construct the most comprehensive stroke neuroimaging dataset of its kind to date. This massive repository of information is a testament to the power of international scientific cooperation.
Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC, underscored the importance of this collaborative approach. "By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge AI, we can detect subtle patterns of brain reorganization that would be invisible in smaller studies," Dr. Toga remarked. "These findings of regionally differential brain aging in chronic stroke could eventually guide personalized rehabilitation strategies." The sheer scale of the data allows for the detection of subtle, nuanced patterns that might otherwise be lost in noise or attributed to individual variability in smaller cohorts. This macro-level perspective is crucial for identifying universal principles of brain adaptation after stroke.
The Dawn of Personalized Stroke Recovery
The implications of this research extend far beyond academic curiosity, pointing towards a future where stroke recovery is more precisely tailored to the individual. The researchers are committed to building upon these foundational discoveries by embarking on longitudinal studies. This next phase will involve tracking patients over time, from the acute stages immediately following a stroke through their long-term recovery trajectories. By meticulously documenting how brain aging patterns and structural changes evolve, clinicians will gain invaluable insights to customize rehabilitation interventions. The ultimate goal is to optimize recovery outcomes, improve functional independence, and enhance the overall quality of life for stroke survivors.
The study’s findings suggest that the brain’s response to stroke is not a monolithic process but rather a complex, dynamic interplay of damage and compensatory adaptation. The identification of "younger" brain regions in contralesional areas, particularly within networks involved in motor control and executive functions, provides a quantifiable marker of this adaptive process. This could potentially lead to the development of new diagnostic tools that assess the brain’s compensatory capacity and predict a patient’s responsiveness to different therapeutic approaches.
For instance, a patient exhibiting strong evidence of contralesional frontoparietal network rejuvenation might be a good candidate for therapies that leverage this adaptive potential, perhaps through targeted cognitive training or neurofeedback techniques designed to further enhance the function of these younger-appearing regions. Conversely, individuals showing less evidence of such compensatory changes might benefit more from interventions focused on restoring function in the damaged hemisphere or exploring alternative motor pathways.
The study’s funding by the National Institutes of Health (NIH) grant R01 NS115845, coupled with the invaluable contributions of international collaborators from institutions including the University of British Columbia, Monash University, Emory University, and the University of Oslo, highlights the global significance of this research. The interdisciplinary nature of the ENIGMA consortium, bringing together neurologists, neuroimaging specialists, computer scientists, and statisticians, was essential for tackling such a complex challenge.
The ability to observe and quantify neuroplasticity through AI-driven brain age estimation represents a significant leap forward. Traditional neuroimaging techniques, while invaluable for identifying structural damage, often fall short in capturing the dynamic functional reorganization that occurs in the brain over time. The brain-PAD metric, derived from deep learning, offers a novel lens through which to view this adaptive process, potentially providing a more sensitive indicator of recovery potential than traditional measures alone.
This research also opens avenues for exploring the molecular and cellular mechanisms underlying this observed "rejuvenation." While the study focuses on structural and functional patterns, future research could investigate whether these younger-appearing brain regions exhibit distinct patterns of gene expression, protein synthesis, or synaptic plasticity that contribute to their enhanced compensatory capacity. Understanding these underlying biological processes could unlock even more targeted therapeutic interventions.
The video resource provided by the Stevens INI, titled "Contralesional Neuroplasticity and Motor Impairment in Chronic Stroke," further illustrates these complex associations, offering a visual and educational companion to the study’s findings. This commitment to public outreach and knowledge dissemination is vital for translating scientific breakthroughs into tangible benefits for patients and healthcare professionals.
In conclusion, the study published in The Lancet Digital Health marks a pivotal moment in stroke research. By harnessing the power of AI and international collaboration, researchers have uncovered a profound insight into the brain’s remarkable capacity for adaptation. The discovery of "younger" brain regions in contralesional hemispheres of stroke survivors with severe motor impairments provides a new paradigm for understanding neuroplasticity and paves the way for more personalized and effective stroke recovery strategies, ultimately aiming to restore function and improve the lives of millions affected by this debilitating condition worldwide.