For decades, the haircare industry has championed "hydration" as the ultimate goal for luscious locks, inundating consumers with products promising to infuse hair with essential moisture. Yet, beneath this marketing narrative lies a complex scientific reality that often contradicts popular belief, leading to widespread confusion among both consumers and seasoned hair experts. The true interaction between water and hair is far more nuanced than a simple "yes" or "no" to hydration, challenging deeply ingrained perceptions and offering a clearer path to genuine hair health.
The "Hydration" Paradox: A Market-Driven Myth?
The concept of "hydrated hair" has become a cornerstone of beauty marketing, suggesting that adding water directly translates to softer, smoother, and healthier-feeling strands. This pervasive belief is so entrenched that, as some cosmetic scientists have revealed, the industry often opts to perpetuate the myth rather than embark on the difficult task of re-educating a public accustomed to simplistic solutions. The commercial imperative to sell products often overshadows the intricate biochemistry of hair, leading to a disconnect between scientific understanding and consumer language. This strategic ambiguity, while commercially effective, contributes to the very confusion it aims to resolve.
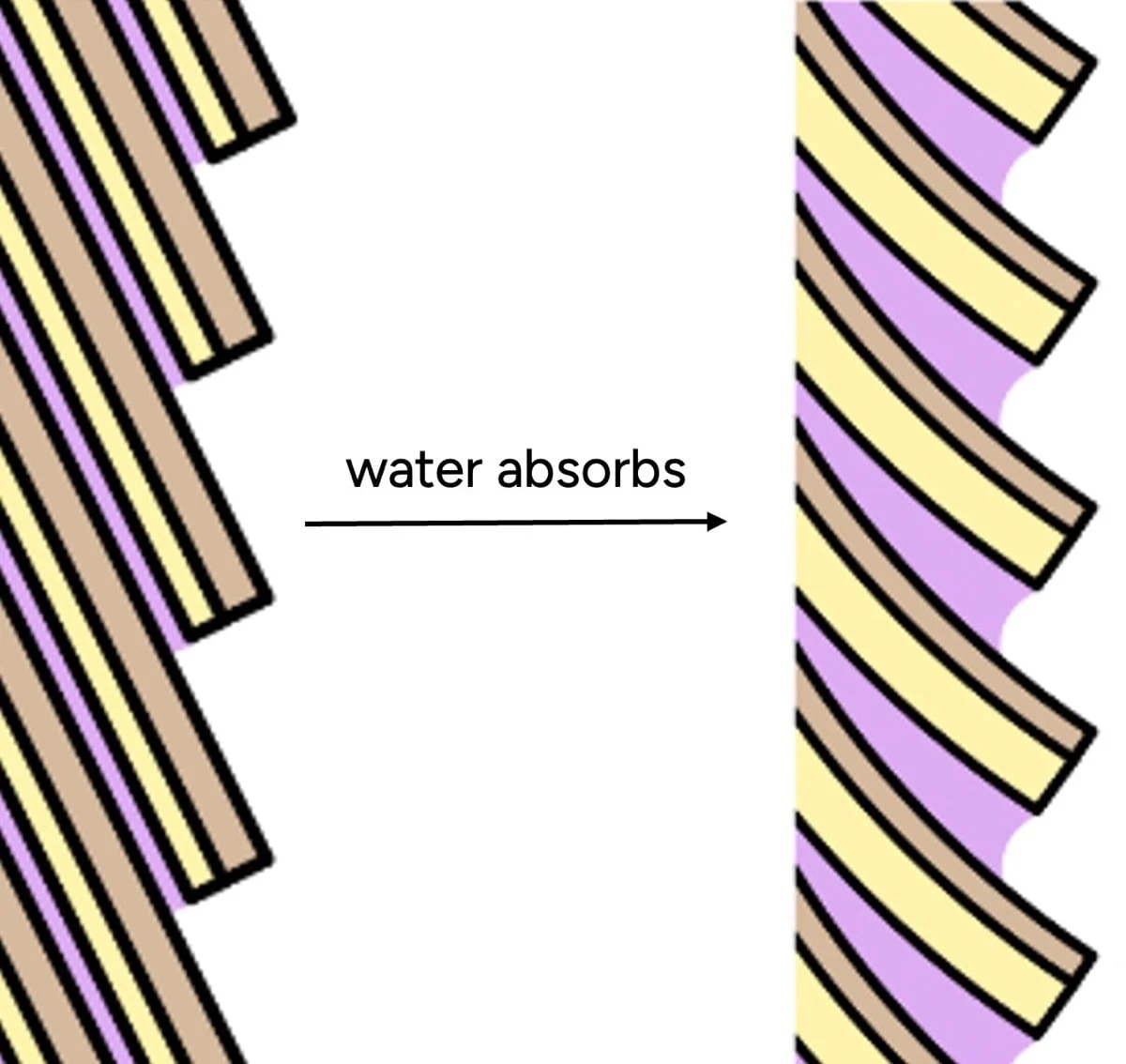
However, a deeper dive into the molecular structure of hair reveals that the conventional understanding of "hydration" is, in many ways, a misnomer. Scientific studies and expert analyses suggest that what consumers perceive as "hydrated" hair — soft, smooth, and manageable — often corresponds to hair with lower intrinsic water content, not higher. This paradox underscores the need to differentiate between the sensory experience of hair and its actual molecular state concerning water.
The Molecular Dance: Water’s Interaction with Hair Bonds
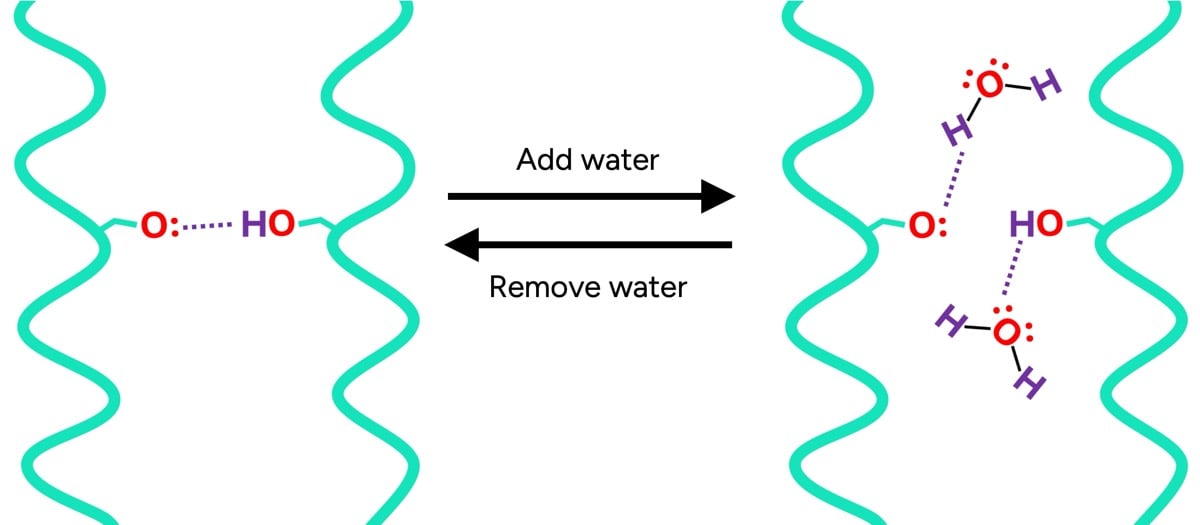
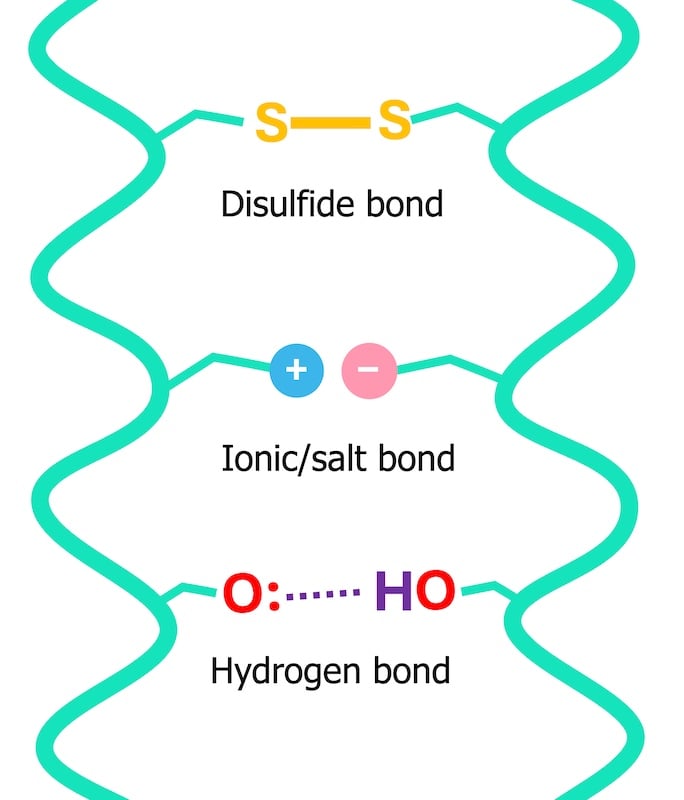
To truly grasp the role of water, one must first understand the fundamental building blocks of hair: proteins. These proteins, primarily keratin, are held together by a network of bonds that dictate hair’s strength, elasticity, and overall shape. While some bonds, like disulfide bonds, are permanent and contribute to hair’s structural integrity (influencing curl patterns), others are temporary, such as hydrogen bonds. These temporary bonds are crucial for hair’s flexibility and ability to hold a style.
The inner layers of the hair shaft, particularly the cortex, are remarkably sensitive to water. In dry hair, temporary bonds form robust connections between adjacent protein chains, akin to molecular "hand-holding." These numerous inter-protein bonds create a relatively rigid and strong structure. However, when water is introduced, its molecules readily form hydrogen bonds with the hair proteins. This creates a competitive scenario: the proteins, now "holding hands" with water molecules, form fewer bonds with each other. The consequence is a reduction in the overall number of inter-protein bonds, rendering wet hair inherently weaker and more elastic.
This phenomenon can be likened to a social gathering: imagine two individuals (hair proteins) who are closely bonded. If a crowd of friendly newcomers (water molecules) enters the room, the original two individuals will naturally spend less time interacting solely with each other as they engage with the new arrivals. Their original bond, while still present, becomes relatively weaker and more flexible within the larger social context. When the "newcomers" (water) eventually leave, the original strong bond between the two individuals (proteins) can reform. This molecular rearrangement is central to how hair responds to wetting and drying.
Detangling Dilemma: Wet vs. Dry Hair Mechanics
The impact of water on hair bonds has significant practical implications, particularly concerning detangling. The reduced internal bonding in wet hair means it is stretchier and more elastic, making it susceptible to damage if handled improperly. For straight hair, the consensus among scientists is that combing it while wet can be more damaging. Although more flexible, the hair strands are weaker, and the presence of water can increase friction between individual strands, leading to greater resistance and breakage during detangling. A study by Epps and Wolfram in 1983 highlighted these distinctions, showing how different environments affect combing force.
Conversely, for curly and coily hair, wet detangling is often less damaging. While the hair is still weaker when wet, the inherent structural complexity of curly hair means that water can help to loosen tangles and reduce the overall resistance encountered during combing. The lubricating effect of conditioner, applied generously in wet conditions, further minimizes friction, allowing for smoother detangling with less force. This highlights a critical point: the optimal detangling method is not universally applicable but depends heavily on hair type and the specific conditions (e.g., presence of conditioner). Even high humidity, which represents a moderate level of water absorption, can alter hair’s mechanical properties, influencing styling and detangling efforts.
| Hair Type | State | Bonds | Strength | Elasticity | Friction | Detangling Damage |
|---|---|---|---|---|---|---|
| Straight | Dry | More | Stronger | Stiffer | Lower | Less |
| Straight | Wet | Fewer | Weaker | Stretchier | Higher | More |
| Curly/Coily | Dry | More | Stronger | Stiffer | Higher | More |
| Curly/Coily | Wet | Fewer | Weaker | Stretchier | Lower | Less |
(Table adapted from original content for journalistic presentation)
Sensory Deception: Why "Dry" Hair Feels "Hydrated"
One of the most counterintuitive findings in hair science is the disconnect between how hair feels and its actual water content. A renowned study, cited by Evans in Cosmetics & Toiletries (2013), asked 50 individuals to evaluate hair samples with varying water content. The overwhelming majority reported that hair with less water felt more moisturized, smoother, less tangled, and less damaged. This phenomenon challenges the very foundation of the "hydration" myth.
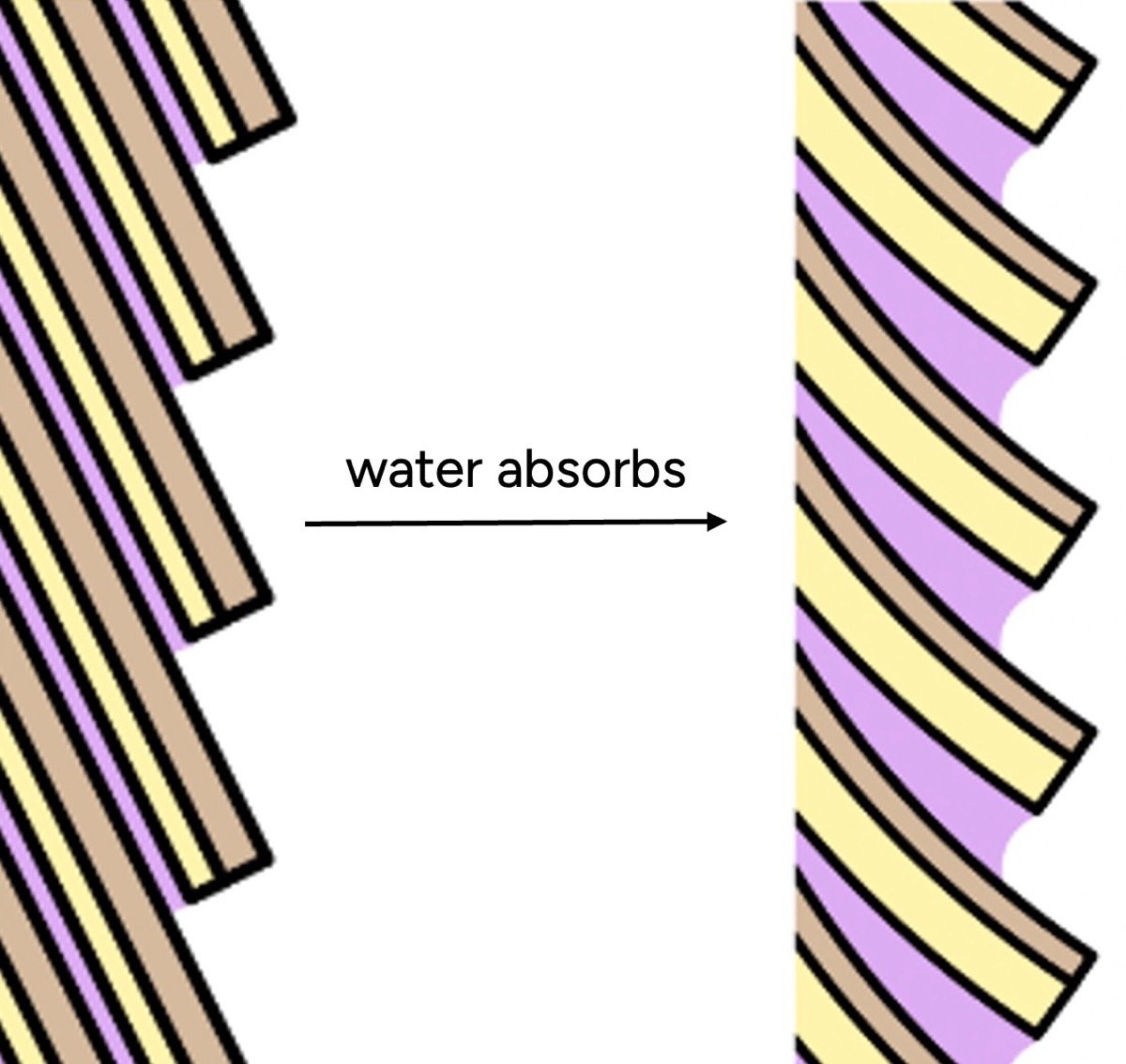
The explanation lies at the microscopic level, specifically with the hair cuticle – the outermost layer composed of overlapping scales. In its dry state, these scales lie relatively flat, creating a smooth surface. However, when hair absorbs water, the cuticle scales swell. Crucially, this swelling is not uniform. The endocuticle (the underside of each scale) and the cell membrane complex (the "glue" between scales) absorb significantly more water than the top surface. This differential swelling causes the scales to lift and buckle, creating a microscopically rough and uneven surface.
Furthermore, water itself possesses inherent stickiness and contributes to friction, a property we exploit when wetting our fingers to get a better grip. Therefore, when hair has a higher water content, its surface becomes bumpier and grippier. Our tactile senses interpret this roughness as "dryness" or damage, contrasting with the smooth, cool sensation we associate with healthy hair. This misinterpretation arises because, unlike most materials we encounter, hair feels rougher and less desirable when wet, leading to a reversal of our typical sensory associations with moisture.
The True Mechanism of "Hydrating" Products
Given the scientific insights, how do "hydrating" hair products actually work? The answer is that they primarily function as conditioners. Conditioners are formulated with emollients and film-forming agents, such as silicones, fatty alcohols, and certain oils. These ingredients deposit a thin, protective layer onto the hair cuticle. This layer serves multiple purposes: it smooths down the lifted cuticle scales, reduces friction between individual strands, and enhances the overall slip and softness of the hair.
Essentially, these products aim to replicate and restore the hair’s natural, protective oily layer, known as the F-layer, which is often lost or damaged through daily wear, chemical treatments, and environmental exposure. By creating an artificial F-layer, conditioners improve the hair’s tactile properties, making it feel smooth, soft, and easy to comb – qualities that consumers associate with "hydration," despite not directly increasing the hair’s internal water content. While some products may include humectants (like glycerin) that attract and hold a small amount of water from the environment, their primary beneficial effect on hair feel is conditioning, not significant internal water absorption.
The Ritual of Washing: Beyond Mere Cleansing
The universal experience of hair feeling "better" after a thorough wash and condition, compared to simply applying a leave-in product, is not primarily about sustained hydration. Instead, it involves a complex interplay of water’s physical and chemical properties that temporarily reset and reshape the hair.
- Enhanced Ingredient Distribution: Water acts as an excellent vehicle for spreading conditioning ingredients evenly across the hair shaft. A rinse-off conditioner, distributed in the presence of ample water, can coat each strand more uniformly, leading to a more consistent smoothing effect.
- Resetting Hair Shape: Water is remarkably effective at breaking the temporary hydrogen bonds that define a hairstyle. This is why washing hair removes heat-styled curls or straightness, and why humidity can quickly ruin a carefully crafted look. By disrupting these bonds, water allows the hair to return to its natural configuration, dictated by its permanent disulfide bonds. This "super floppy" state, facilitated by warm water and shampoo (which reduces surface tension), makes hair highly malleable and receptive to new styling.
- Clump Formation in Textured Hair: For curly and coily hair, water plays an additional crucial role in facilitating strand alignment and curl definition. Due to surface tension, water coats the hair strands and makes them slightly sticky. When wet strands come into contact, water can "zip" them together through a process called self-assembly, forming distinct curl clumps. These larger, aligned clumps can create the illusion of "hydration" even when dry. A smooth, aligned surface transfers heat more efficiently from the hand, making it feel cooler and smoother – a sensation our brains often interpret as "moisturized," similar to how satin feels cooler than rough cotton.
- Microscopic Cuticle Reset: Rapid or uneven drying, particularly with hot tools, can cause the cuticle scales to dry at different rates, leading to buckling and an irregular hair surface (as documented by Gamez-Garcia, 2021). Soaking hair in water and allowing it to dry naturally can help reset these buckled scales, returning the cuticle to a flatter, smoother configuration. This microscopic reshaping contributes to the overall improved feel and appearance of freshly washed and dried hair.
Crucially, none of these effects necessitate a permanently elevated water content within the hair. Instead, they demonstrate how water acts as a transient agent, entering the hair to facilitate changes in structure and distribution of products, and then leaving, allowing the hair to settle into a desired state.
The Goldilocks Zone: Optimal Water Content for Hair Health

Just like skin, hair thrives within an optimal range of water content – a "sweet spot" that is neither too wet nor too dry. While continuous high water content can lead to fragility and a rough feel, too little water also poses significant challenges for hair health.
Hair that is excessively dry exhibits several negative characteristics:
- Increased Static Build-up: In very low humidity environments, hair is prone to static electricity. This occurs because dry hair readily loses electrons when rubbed against other materials, and without sufficient water molecules to dissipate these charges, static builds up, causing flyaways and frizz.
- Brittleness and Reduced Shock Absorbency: Severely dry hair becomes stiff and brittle. The abundance of temporary bonds, unopposed by water molecules, makes the hair less flexible. This reduced flexibility means hair is less able to absorb mechanical stress from actions like brushing or styling. Instead of bending, it can form microscopic cracks that propagate, eventually leading to breakage. The analogy with fingernails, made of similar proteins, is apt: dry nails splinter and crack when clipped, while wet nails are more flexible and cut cleanly.
The ideal water content allows hair to be flexible enough to withstand daily manipulation without cracking, yet firm enough to maintain its structure and feel smooth. Achieving this balance involves managing how water interacts with hair, rather than simply drenching it.
Practical Applications and Future Directions
Understanding the true science of hair, hydration, and water empowers consumers to make more informed haircare choices. The key takeaways are clear:
- Water is a Tool: Water itself is neither inherently good nor bad for hair. It’s a powerful tool that temporarily alters hair’s structure and flexibility, which can be leveraged for styling or lead to damage if mishandled. The concept of "hygral fatigue" – the idea that repeated wetting and drying inherently damages hair – is largely unsubstantiated.
- Prioritize Conditioning: For hair that feels smooth, soft, and healthy, focus on effective conditioning. Products that condition, rather than simply claim to "hydrate," are essential for smoothing the cuticle, reducing friction, and improving tactile feel. The specific terminology ("hydrating") on a product label is less important than its functional ingredients (emollients, film-formers).
- Gentle Wet Hair Care: Always treat wet hair with extra care. Since it is weaker, minimize aggressive brushing or styling. Utilize ample conditioner to provide slip and reduce friction, especially during detangling, regardless of whether hair is wet or dry.
- Manage Environmental Humidity: Be aware of how ambient humidity affects your hair’s properties and adjust your routine accordingly. In very dry conditions, products that help manage static can be beneficial.
The cosmetic science community continues to bridge the gap between complex molecular realities and consumer expectations. This ongoing effort aims not just to develop more effective products but also to educate the public, fostering a deeper, more accurate understanding of hair health beyond marketing buzzwords. By appreciating the intricate dance between water and hair, individuals can cultivate routines that truly nourish and protect their strands, moving beyond myths to embrace evidence-based haircare.
References
Evans T. Consumer vs. scientific language: relating in vivo to in vitro. Cosmetics & Toiletries. 2013;128(5):300-304.
Epps J, Wolfram LJ. Letter to the editor. J Soc Cosmet Chem. 1983;34:213-214.
Gamez-Garcia M. Moisture in the cuticle sheath: effects on hair mechanical and cosmetic properties. J Cosmet Sci. 2021;72:687-696.
Kamath YK, Hornby SB. Effect of chemical and humectant treatments on the mechanical and fractographic behavior of Negroid hair. J Soc Cosmet Chem. 1985;36:39-52.
Kaushik V, Chogale R, Mhaskar S. Single hair fiber assessment techniques to discriminate between mineral oil and coconut oil effect on hair physical properties. J Cosmet Dermatol. 2021;20(4):1306-1317. doi:10.1111/jocd.13724
Robbins CR. Chemical and Physical Behavior of Human Hair. 5th ed. Springer Berlin Heidelberg 2012.
Wong M. Hair, hydration and water: the real science. Lab Muffin Beauty Science. January 28, 2026. Accessed March 11, 2026. https://labmuffin.com/hair-hydration-and-water-the-real-science/