A groundbreaking new study, published in the prestigious journal The Lancet Digital Health, has unveiled a surprising and potentially revolutionary insight into how the human brain responds to the devastating effects of stroke. Researchers at the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have discovered that individuals experiencing severe physical impairments following a stroke may exhibit characteristics of a "younger" brain structure in areas that remained unaffected by the initial injury. This remarkable adaptation appears to be a testament to the brain’s inherent capacity for reorganization and repair in the face of significant trauma.
The ambitious research initiative was conducted under the umbrella of the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, a global consortium dedicated to unraveling the complexities of stroke recovery. This comprehensive study involved the meticulous analysis of brain scans from an impressive cohort of over 500 stroke survivors. These valuable data were collected from 34 distinct research centers spanning eight different countries, creating an unparalleled international dataset. To process and interpret this vast amount of information, the team employed sophisticated deep learning models, which had been rigorously trained on tens of thousands of magnetic resonance imaging (MRI) scans. This advanced artificial intelligence approach enabled researchers to accurately estimate the "brain age" of various regions within each hemisphere of the brain, thereby providing a novel perspective on how stroke impacts both brain structure and the subsequent recovery process.
AI Uncovers the Brain’s Rewiring Mechanism Post-Stroke
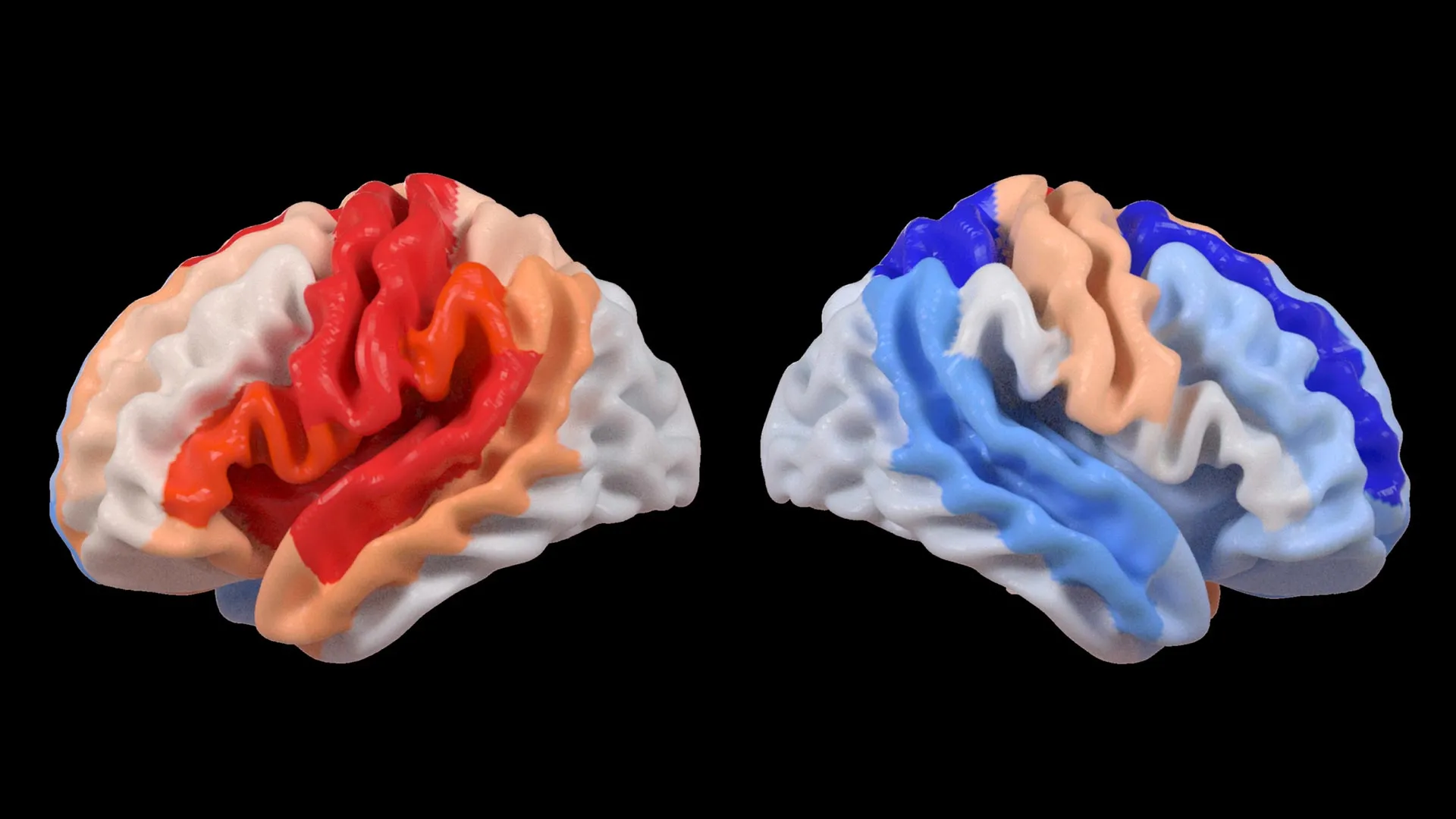
At the core of this investigation was the application of a cutting-edge artificial intelligence technique known as a graph convolutional network. This sophisticated system was instrumental in estimating the biological age of 18 specific brain regions by analyzing their intricate structural patterns depicted in the MRI data. The researchers then meticulously compared this AI-estimated biological age to each participant’s chronological age. The resulting metric, termed the brain-predicted age difference (brain-PAD), serves as a crucial indicator of overall brain health and integrity. A significant positive brain-PAD suggests that a brain region appears older than its chronological age, often indicative of damage or decline, while a negative brain-PAD suggests a region appears younger than its chronological age.
The findings from this analysis revealed a striking correlation when these brain age measurements were juxtaposed with standardized motor function scores. Stroke survivors who presented with severe limitations in their physical movement, even after enduring more than six months of intensive rehabilitation, displayed a pattern of younger-than-expected brain age in regions located on the side of the brain opposite to the original stroke lesion. This phenomenon was particularly pronounced within the frontoparietal network, a critical neural circuitry involved in a complex array of functions, including the planning of movements, the allocation of attention, and the coordination of actions.
Dr. Hosung Kim, an associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author of the study, elaborated on these remarkable observations. "We discovered that larger strokes tend to accelerate the aging process in the hemisphere that was directly damaged," Dr. Kim stated. "However, paradoxically, we observed that the opposite side of the brain appeared younger. This distinct pattern strongly suggests that the brain is actively reorganizing itself, essentially rejuvenating undamaged neural networks to compensate for the lost functionality resulting from the injury."
This apparent rejuvenation in undamaged brain regions, particularly the contralesional frontoparietal network, indicates a profound form of neuroplasticity. This network, known for its role in supporting motor planning, attention, and coordination, exhibiting a more "youthful" structural signature in severely impaired individuals, suggests an active compensatory mechanism. Instead of simply reflecting a complete restoration of motor function, this pattern appears to represent the brain’s ingenious attempt to adapt and function when the primary motor system has been compromised and can no longer operate optimally. This provides researchers with an unprecedented visual representation of neuroplasticity, offering insights that traditional imaging techniques might have previously obscured.
Leveraging Global Data to Illuminate Hidden Neural Patterns
The immense success and depth of this study were significantly amplified by its reliance on the ENIGMA initiative. ENIGMA represents a monumental global collaboration, pooling data and expertise from over 50 countries to foster a more profound understanding of the human brain across a spectrum of neurological conditions. By establishing standardized protocols for MRI data acquisition and the collection of clinical information from a multitude of research groups, the team successfully curated the most extensive stroke neuroimaging dataset of its kind ever assembled.
Arthur W. Toga, PhD, the director of the Stevens INI and a Provost Professor at USC, emphasized the power of this large-scale data aggregation. "By pooling data from hundreds of stroke survivors worldwide and applying cutting-edge artificial intelligence, we are able to detect subtle patterns of brain reorganization that would likely remain invisible in smaller, more localized studies," Dr. Toga remarked. "These findings, particularly the regionally differential brain aging observed in chronic stroke patients, hold immense promise for guiding the development of more personalized and effective rehabilitation strategies in the future."
The genesis of this research can be traced back to the ongoing efforts within the ENIGMA consortium, which has a history of leveraging large, harmonized datasets to address complex neuroscientific questions. The stroke recovery working group, specifically, has been actively collecting and standardizing data for several years, aiming to identify biomarkers and predictors of recovery. The current study represents a significant leap forward by integrating advanced AI methodologies with this rich, multi-site dataset.
The timeline of this research involved several key phases. Initial data collection and harmonization under the ENIGMA framework likely began several years prior. The development and training of the deep learning models would have been an intensive process, followed by the application of these models to the stroke survivor cohort. The subsequent analysis and correlation with clinical data, including motor function scores, formed the crucial interpretative stage, culminating in the publication of the findings in The Lancet Digital Health.
Charting a Course Towards Personalized Stroke Rehabilitation
Looking ahead, the researchers are committed to advancing this critical line of inquiry. Their future plans include longitudinal studies, where they will meticulously track patients over extended periods, from the immediate aftermath of a stroke through the various stages of long-term recovery. By continuously monitoring how brain aging patterns and structural changes evolve over time, scientists aim to gain a deeper understanding of the dynamic nature of brain adaptation. This invaluable knowledge is expected to empower clinicians to tailor rehabilitation treatments with unprecedented precision, aligning interventions with each individual’s unique recovery trajectory. The ultimate goal is to significantly enhance patient outcomes and elevate their overall quality of life.
The implications of these findings extend far beyond the immediate scientific community. For patients and their families, this research offers a beacon of hope, suggesting that the brain possesses a remarkable, albeit complex, capacity to adapt and compensate. For clinicians and therapists, it provides new avenues for assessing recovery and potentially for designing more targeted and effective interventions. The identification of specific brain regions that exhibit "youthful" patterns in the presence of severe motor deficits could lead to the development of novel therapeutic strategies aimed at stimulating or supporting these compensatory networks.
The study’s reliance on a diverse international cohort is also a crucial aspect, highlighting the universal nature of brain plasticity. While stroke is a global health challenge, affecting millions worldwide each year—with an estimated 15 million new strokes occurring annually according to the World Health Organization—individual recovery pathways can vary significantly. This research underscores the importance of a global, collaborative approach to understanding and addressing this complex condition.
The National Institutes of Health (NIH) has recognized the significance of this work by providing funding through grant R01 NS115845. This support underscores the potential of this research to make a substantial impact on stroke neuroscience. International collaborators, including institutions such as the University of British Columbia, Monash University, Emory University, and the University of Oslo, have played a vital role in contributing data and expertise, underscoring the collaborative spirit that drives major scientific breakthroughs.
The potential for personalized rehabilitation is a particularly exciting implication. Traditionally, stroke rehabilitation has often followed generalized protocols. However, this study’s findings suggest that by understanding the specific patterns of brain reorganization, therapists could potentially:
- Identify individuals who might benefit most from specific types of therapy: For instance, those showing strong compensatory activity in the frontoparietal network might respond better to attention-focused or planning-oriented exercises.
- Develop novel neurofeedback or stimulation techniques: Targeting the "younger" appearing contralesional regions could potentially enhance their compensatory role.
- Predict long-term recovery potential: While the study focuses on chronic stroke, understanding these early compensatory mechanisms could offer insights into long-term prognoses.
The study’s methodology, employing deep learning on a massive, harmonized dataset, represents a paradigm shift in neuroimaging research. This approach allows for the detection of subtle, complex patterns that are often missed by traditional statistical methods or smaller sample sizes. As Dr. Toga noted, "These are patterns that would be invisible in smaller studies." This highlights the power of big data and advanced analytics in unlocking new biological insights.
The video linked within the study, created by the Stevens INI, offers a visual exploration of the associations between contralesional neuroplasticity and motor impairment, providing a valuable educational resource for both the scientific community and the public. Such supplementary materials are crucial for disseminating complex scientific findings effectively and fostering broader understanding and engagement.
In conclusion, this research from the USC Stevens INI, in collaboration with the global ENIGMA consortium, marks a significant advancement in our understanding of the brain’s remarkable resilience and adaptability after stroke. The discovery of "younger" appearing brain structures in undamaged regions, particularly within the frontoparietal network, in individuals with severe motor impairments, offers a compelling new perspective on neuroplasticity. This finding not only deepens our fundamental knowledge of brain function but also paves the way for the development of more personalized and effective stroke rehabilitation strategies, ultimately aiming to improve the lives of millions affected by this debilitating condition worldwide. The ongoing longitudinal studies will be crucial in translating these exciting insights into tangible clinical benefits.