A widely circulated beauty hack purporting to test the efficacy of heat protectants using thermal paper receipts has been thoroughly debunked by scientific investigation, revealing that the viral method offers misleading results that have no bearing on a product’s ability to safeguard hair. The popular test, which involves spraying a heat protectant onto a thermal paper receipt and then applying a flat iron hair straightener, gained traction on social media platforms like TikTok, with proponents claiming that receipts that remain white indicate more effective heat protection. However, a detailed inquiry by Lab Muffin Beauty Science, led by chemist Michelle Wong, has uncovered that the observed whitening effect is primarily due to the solvent properties of the products and the cooling effect of residual moisture, rather than genuine heat protection.

The Rise of a Viral Phenomenon and Initial Plausibility
The thermal paper test emerged as a compelling visual demonstration within the burgeoning landscape of social media beauty content. Content creators, including cosmetologist Lucy Seitz, systematically applied various heat protectants to thermal receipts, documenting the immediate reaction when a hot flat iron passed over the treated paper. Since thermal paper is designed to turn black when exposed to heat, the logic posited that areas remaining white were effectively insulated, thus indicating superior heat protection. The test’s appeal lay in its apparent simplicity, visual clarity, and the seemingly direct analogy: if heat damages hair and makes receipts black, then preventing the receipt from blackening should equate to hair protection.

In scientific methodology, testing on "models" – substances or systems that mimic the characteristics of the real thing – is a common practice. For instance, pharmaceuticals are often tested on mice, and skincare products on reconstituted human skin models like L’Oréal’s EpiSkin, which has significantly reduced the need for animal testing. The receipt test, at first glance, seemed to offer a similar model: a heat-sensitive substrate (thermal paper) interacting with heat-protective agents, much like hair interacts with styling tools. This "surface validity" initially gave the viral test an air of credibility, prompting a deeper scientific dive into its underlying mechanisms.
The Scientific Inquiry Begins: Replicating and Observing

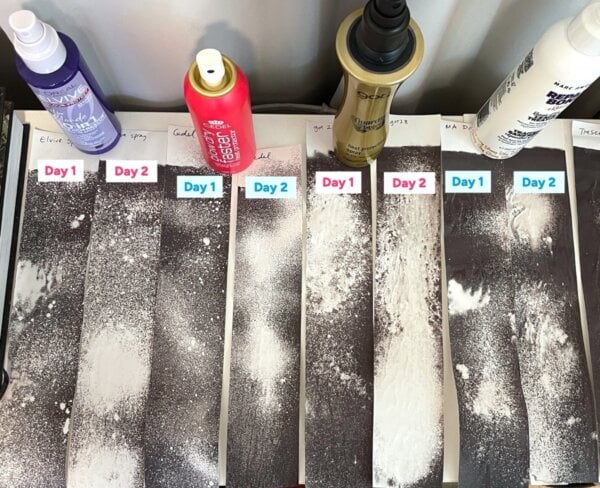
Michelle Wong of Lab Muffin Beauty Science embarked on a comprehensive investigation to ascertain the validity of this viral claim. Her initial experiments aimed to replicate the conditions observed in popular videos. She gathered 11 products with heat protection claims, encompassing a variety of formulations: seven pump sprays, one propellant spray, and three cream products. Using a flat iron set to 170°C (approximately 340°F), a temperature comparable to that used in the viral demonstrations, Wong began her tests.
An immediate and critical observation was made: when heat protectant was sprayed onto a receipt and immediately subjected to the flat iron, it often crackled and smoked. This indicated the presence of moisture. Wong adjusted her protocol, allowing products to dry for about 15 minutes, mimicking a more realistic application scenario where products might be allowed some time to settle.

The initial results from these replicated tests revealed a significant impact of drying time. Receipts tested immediately after application showed drastically different results compared to those allowed 15 minutes to dry, underscoring the influence of residual liquid. Cream products consistently resulted in lighter receipts compared to sprays. This was attributed to the generally thicker application of creams, providing more insulation, and their slower drying times, which meant more residual water was likely present. Conversely, two sprays – Goldwell and IGK – which had water lower on their ingredient lists, yielded the darkest receipts, suggesting less water content and thus less heat buffering. These preliminary findings pointed towards water content and application thickness as significant variables, confounding any direct correlation to actual heat protectant efficacy.
The Critical Role of Water and the "Bubble Hair" Phenomenon

Building on the initial observations, the investigation then focused on the cooling effect of water. Water possesses a high specific heat capacity and latent heat of vaporization, meaning it can absorb a substantial amount of thermal energy before its temperature rises significantly or it evaporates. This property makes water an effective, albeit problematic, cooling agent.
Wong conducted dedicated water tests, comparing receipts that were either "dunked" in water or "sprayed" with water, then subjected to the flat iron after varying drying times (0, 2, 5, and 10 minutes). Dunked receipts consistently remained white for longer durations, demonstrating water’s strong cooling capability. Sprayed receipts, however, showed a more nuanced effect: while immediately sprayed receipts had some white areas, they turned progressively greyer and then blacker as drying time increased. This confirmed that water’s presence significantly influenced the thermal paper’s reaction, with its cooling effect diminishing as it evaporated.

Crucially, while water effectively cools the receipt, it is a detrimental component when using hot tools on hair. When wet hair is exposed to high heat from a flat iron, the water inside the hair shaft can explosively boil and evaporate, leading to the formation of "bubble hair." This phenomenon causes severe structural damage to the hair cuticle and cortex, weakening the hair and making it more prone to breakage. Therefore, products that keep receipts white primarily due to their water content are not indicating effective heat protection for hair; rather, they are demonstrating a mechanism that could potentially cause more damage to hair if not thoroughly dried before styling. The fundamental purpose of a heat protectant is not to block heat entirely, but to distribute it evenly and create a barrier that reduces direct thermal damage and friction, not to cool the hair by evaporation.
An Unforeseen Complication: The True Nature of Thermal Paper

A pivotal moment in the investigation came with a re-evaluation of thermal paper’s heat sensitivity. Initial Google searches for "thermal paper temperature" had yielded a range of 150-185°C, seemingly aligning with hair straightener temperatures. However, a deeper dive into the obscure chemistry of thermal paper, including a German Wikipedia article and specialized industry data, revealed a significant discrepancy. The actual temperature at which thermal paper begins to develop color is much lower, typically between 60 to 100°C (140-212°F), and reaches applicable density between 70 and 120°C (158-248°F).
This discovery was a "huge spanner in the works," as Wong described it. Hair damage, particularly protein denaturation and lipid degradation, typically begins around 100°C. If receipts were reacting at temperatures significantly lower than this, the entire model would be invalidated, as it would be indicating "protection" long before hair would even begin to suffer damage.

To resolve this, Wong conducted careful temperature tests using various receipts immersed in cooling boiling water. These experiments definitively showed that most receipts turned black around 95°C. This finding was crucial, as it confirmed that thermal paper’s activation temperature does align with the threshold for hair damage, suggesting that the model wasn’t entirely flawed in its heat sensitivity. However, this still did not explain the perplexing "Day 2" results, where thoroughly dried products often resulted in lighter receipts, contradicting the expectation that fully evaporated water would lead to darker marks.
Unraveling the Mechanism: The Hypothesis of Solvent Action

The persistent observations of receipts turning grey before heating, or becoming lighter after 24 hours of drying, could not be explained by water’s cooling effect alone. This led to a new hypothesis: the "effectiveness" of certain heat protectants in this test was not due to heat protection, but rather to their ability to dissolve or disrupt the ink layer on the thermal paper.
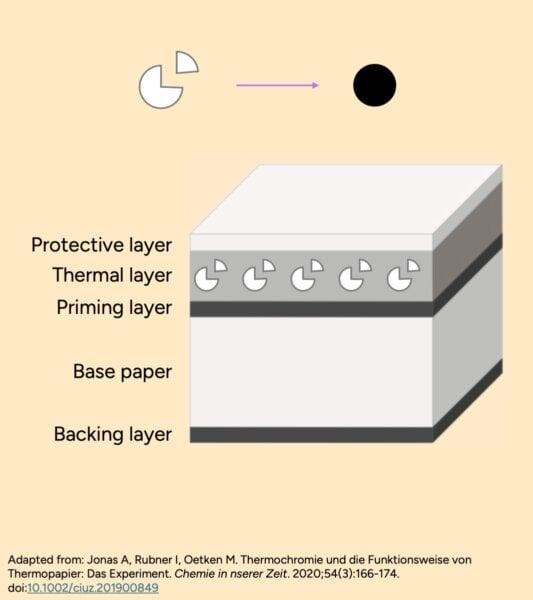
Thermal paper functions through a complex chemical reaction. It consists of a top layer containing invisible dye precursors, a developer, and a sensitizer embedded within a solid matrix, often a wax-like substance. When heated, this matrix melts, allowing the dye and developer to mix and react, forming a visible black image. The ink layer is delicate, and its formation is dependent on specific conditions.

Wong noticed that many of the "effective" sprays contained high amounts of alcohol. Alcohol is a powerful solvent, capable of dissolving a wider range of substances, including the oily components of thermal ink, compared to water. Her revised hypothesis proposed that products containing solvents (like alcohol) or solubilizing agents (like surfactants) were interfering with the thermal ink’s ability to react and form a dark image, or even actively erasing it.
Confirmatory Experiments and Chemical Explanations
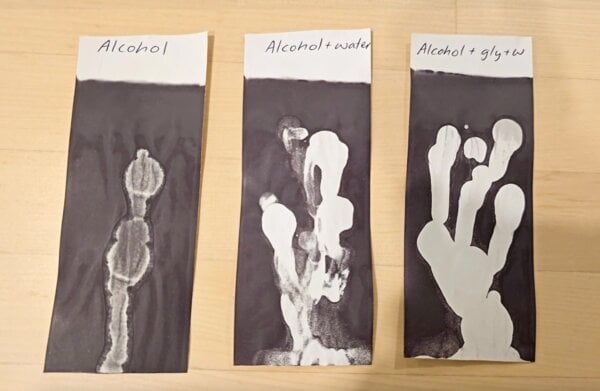
To test this new hypothesis, a series of confirmatory experiments were conducted using substances known for their solvent or solubilizing properties, but which are not hair heat protectants.
-
Alcohol, Water, and Glycerin: Drops of methylated spirits (alcohol), alcohol with water, and alcohol with water and glycerin were applied to receipts. All these combinations kept the receipts light after heating, even after 30 minutes of drying. Interestingly, diluted alcohol proved more effective at whitening than pure alcohol. This is because water and glycerin can form hydrogen bonds with alcohol, slowing its evaporation and allowing it to remain on the receipt for longer, thereby extending its ink-dissolving action. This also explained the puzzling "Day 2" results: the solvent action continued to disrupt the ink over a longer period, resulting in even lighter receipts. Pure water and glycerin, lacking the solvent properties for the oily thermal ink, did not significantly disturb the color change.

-
Other Alcohol-Containing Products: Perfume, which is high in alcohol, immediately turned receipts grey upon application and kept them white after heating. Dry shampoo, also containing alcohol, produced immediate greying but offered less "protection" after heating, likely due to its faster alcohol evaporation and lower humectant content, limiting the duration of solvent action.
-
Surfactants: Many heat protectants, even those without high alcohol content, contain surfactants (emulsifiers) to blend oil and water components. Surfactants are excellent solubilizers, capable of dissolving oily substances. Tests with micellar water and water mixed with detergent demonstrated that these solutions effectively kept receipts white after application and heating. Similarly, common beauty creams like CeraVe Moisturising Cream and Laneige Cream Skin, which contain emulsifiers, also prevented the receipts from darkening, with physical rubbing enhancing the disruption of the ink layer. Sunscreens, often formulated with both alcohol and surfactants, likewise rendered the receipts white.

-
Volatile Silicones: Cyclopentasiloxane, a volatile silicone often found in hair products, had minimal impact on the receipts. This is because it is too oily to readily dissolve the ink layer and evaporates quickly, explaining why some silicone-based heat protectants, which are highly effective on hair, "failed" the receipt test.
-
Propellants: The investigation also explored the role of propellants like HFC-152a, used in some aerosol sprays. However, tests with compressed air (primarily propellant) showed no effect on the receipts, ruling it out as a factor.

-
Erasing Pre-formed Ink: A conclusive experiment involved drawing on thermal receipts with a hot flat iron to create black marks, then applying the solvent-containing substances (alcohol, perfume, sunscreen, micellar water, moisturizer) to these marks. These substances largely succeeded in erasing the black ink, providing strong evidence that they actively dissolve or disrupt the ink layer.
Broader Implications for Consumers and the Beauty Industry

The comprehensive findings unequivocally demonstrate that the viral thermal paper heat protectant test is scientifically invalid. It primarily measures the solvent or solubilizing capabilities of a product’s ingredients on thermal ink, rather than its ability to protect hair from heat damage. This widespread misinformation carries significant implications for consumers and the beauty industry.
Consumers are susceptible to making misinformed purchasing decisions based on visually compelling but scientifically flawed demonstrations. Products that "pass" this test may offer no real heat protection for hair, while genuinely effective products might "fail" simply because their formulation does not contain strong solvents or surfactants that interact with thermal paper ink. This erodes consumer trust and can lead to unnecessary hair damage for those relying on the viral test’s conclusions.
For beauty brands, such viral tests pose a reputational challenge. Products rigorously tested on actual hair using established scientific methodologies might be unfairly maligned on social media. Brands like Amika, whose product "failed" the receipt test, have had to counter the misinformation by showcasing footage of their extensive, instrumental hair testing, involving methods like repeatedly combing treated hair samples in specialized machines to assess damage. This highlights the chasm between superficial viral tests and genuine scientific validation.
Heat protectants do not work by "blocking" heat. Their primary mechanisms involve forming a protective barrier (often with silicones and polymers) that reduces friction, helps distribute heat more evenly along the hair shaft, and prevents hot spots. Some ingredients, like hydrolyzed proteins, can also help reinforce the hair structure. The presence of water in a heat protectant can be beneficial if it’s allowed to evaporate before hot tool use, as it helps distribute the product and penetrate the hair. However, styling wet or damp hair with a hot tool is detrimental due to the "bubble hair" effect.
For consumers seeking effective heat protection, it is paramount to look beyond viral demonstrations and rely on scientifically validated information. Seek out products from brands that explicitly state their testing methodologies, such as "instrumental tests" or comparisons "vs. non-conditioning shampoo alone," often accompanied by specific temperature claims. Key effective ingredients to look for include various silicones (e.g., dimethicone, amodimethicone), film-forming polymers (e.g., PVP/VA copolymer), and hydrolyzed proteins. Crucially, always ensure hair is completely dry before using hot styling tools, even when applying water-based heat protectants, to prevent thermal damage from explosive water evaporation.
In conclusion, while the viral thermal paper test provided an engaging visual, it utterly failed as a scientific model for heat protectant efficacy. The investigation by Lab Muffin Beauty Science underscores the importance of critical thinking and scientific rigor in an age dominated by easily shareable, yet often misleading, online content. True hair protection stems from understanding the science behind product formulations and adhering to proper application techniques, not from the deceptive reactions of a thermal receipt.
