Major depressive disorder (MDD) stands as a pervasive global health crisis, casting a long shadow over millions and ranking among the foremost contributors to worldwide disability. While a significant portion of individuals grappling with depression find relief through conventional antidepressant medications, a disheartening reality persists for approximately 30% who develop treatment-resistant depression (TRD). For these individuals, standard pharmacological interventions prove insufficient, leaving their debilitating symptoms largely unaddressed. In recent years, ketamine has emerged as a beacon of hope, demonstrating remarkable speed and efficacy in alleviating depressive symptoms for those with TRD. However, the precise molecular mechanisms by which ketamine exerts its profound influence within the human brain have remained a significant enigma, hindering efforts to refine and personalize this potentially life-changing therapeutic approach.
A groundbreaking study, published on March 5, 2026, in the esteemed journal Molecular Psychiatry, has taken a monumental step towards demystifying ketamine’s action. Spearheaded by Professor Takuya Takahashi and his dedicated team at the Department of Physiology within Yokohama City University Graduate School of Medicine in Japan, the research employed a sophisticated positron emission tomography (PET) imaging technique. This advanced methodology allowed scientists to directly visualize dynamic changes in the glutamate $alpha$-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor (AMPAR). AMPARs are pivotal proteins that orchestrate communication between neurons, playing a critical role in synaptic plasticity – the brain’s ability to adapt and change – and glutamatergic signaling, a vital neurotransmitter pathway implicated in the therapeutic effects of ketamine in TRD patients.
"Although ketamine has shown rapid antidepressant effects in patients with treatment-resistant depression, its molecular mechanism in the human brain has remained unclear," Professor Takahashi stated, underscoring the long-standing gap in scientific understanding that his team sought to bridge. "Our research represents the first direct visualization of how ketamine influences these crucial brain receptors in living humans."
Visualizing Brain Receptors With a Novel PET Tracer
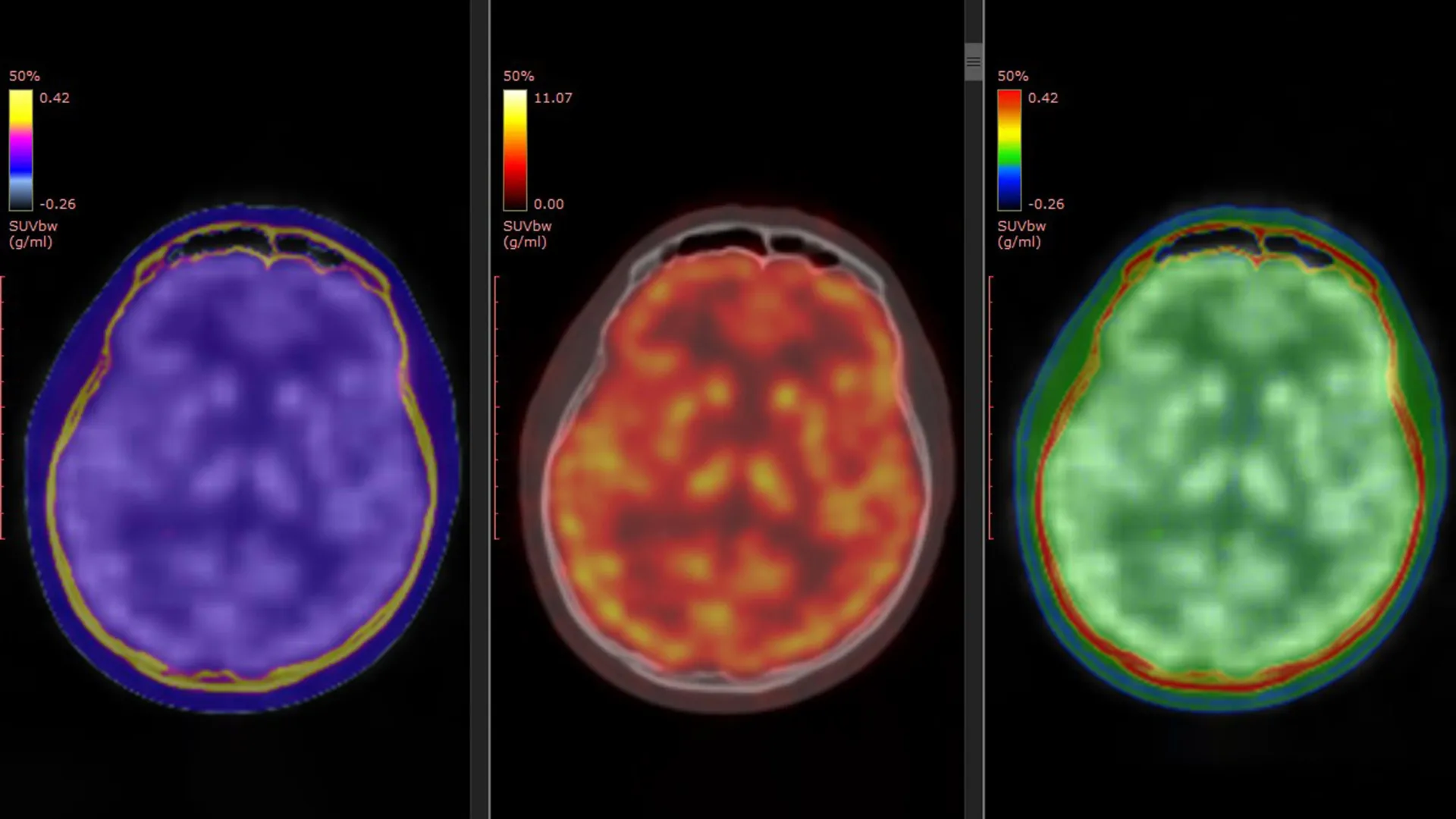
The success of this pivotal study hinges on a novel PET tracer, meticulously developed by Professor Takahashi’s group, designated as [11C]K-2. This innovative tracer possesses the unique capability to illuminate cell-surface AMPARs directly within the living human brain. Prior preclinical research, conducted in laboratory settings and on animal models, had strongly suggested that ketamine’s antidepressant effects were intrinsically linked to AMPAR activity. However, this new investigation provides the first direct, irrefutable evidence of this process occurring within the human brain, validating years of theoretical and experimental work.
To achieve this remarkable feat, the researchers meticulously integrated data from three separate, registered clinical trials conducted within Japan. The collective study cohort comprised 34 individuals diagnosed with treatment-resistant depression and a control group of 49 healthy participants, carefully selected to ensure robust comparative analysis. Over a two-week period, the TRD patients were administered either intravenous ketamine or a placebo in a randomized, controlled fashion. Crucially, PET brain imaging was performed both before the commencement of the treatment regimen and again following the final infusion. This dual-imaging approach enabled the research team to precisely track and quantify any alterations in AMPAR levels and their spatial distribution throughout the brain over the course of the intervention.
Region-Specific Brain Changes Linked to Symptom Relief
The findings of the study revealed striking insights into the neurological underpinnings of ketamine’s efficacy. Individuals diagnosed with TRD exhibited widespread abnormalities in AMPAR density when compared to their healthy counterparts. These discrepancies were not generalized across the entire brain but were notably concentrated within specific neural circuits, suggesting a targeted rather than diffuse impact of the disorder.
Importantly, ketamine’s influence on AMPARs was not uniform across all brain regions. Instead, the observed improvements in depressive symptoms were intrinsically linked to dynamic, region-specific adjustments in AMPAR levels. In certain cortical areas, researchers noted an increase in receptor density, a finding that may correlate with enhanced neural communication and processing. Conversely, in regions associated with reward processing, particularly the habenula – a brain structure known to play a role in mood and motivation – reductions in AMPAR density were observed. These nuanced, region-specific shifts in AMPAR distribution were found to be strongly correlated with the degree of symptom relief experienced by the patients.
"Ketamine’s antidepressant effect in patients with TRD is mediated by dynamic changes in AMPAR in the living human brain," Professor Takahashi elaborated. "Using a novel PET tracer, [11C]K-2, we were able to visualize how ketamine alters AMPAR distribution across specific brain regions and how these changes correlate with improvements in depressive symptoms." These direct human observations lend powerful support to mechanisms previously elucidated in animal studies and, crucially, establish a concrete link between these molecular alterations and tangible clinical antidepressant effects.
Potential Biomarker for Predicting Treatment Response
Beyond clarifying the intricate molecular pathways of ketamine’s action, these findings hold significant promise for practical clinical application. The ability to visualize AMPARs using PET imaging could evolve into a valuable biomarker for predicting how individuals with TRD will respond to ketamine treatment. The persistent challenge in mental health care, particularly for those with TRD, is the identification of reliable biological markers that can accurately forecast treatment outcomes. Many patients who do not achieve remission with standard antidepressants often embark on a difficult and frustrating trial-and-error process with various medications. The development of a predictive biomarker could streamline this process, allowing clinicians to more confidently select the most effective treatment for each individual from the outset.
The implications of such a biomarker are profound. It could enable clinicians to preemptively identify patients who are most likely to benefit from ketamine therapy, thereby avoiding unnecessary exposure to treatments that are unlikely to be effective. Furthermore, it could help in determining the optimal dosage and duration of ketamine treatment, further personalizing the therapeutic approach.
Toward More Personalized Depression Treatments
This pioneering research effectively bridges a long-standing chasm between fundamental laboratory discoveries and their translation into clinical psychiatric practice. By providing the capability to directly observe AMPAR activity in the living human brain, the study unequivocally establishes AMPAR modulation as a central mechanism driving ketamine’s rapid antidepressant effects. This insight paves the way for the development of more targeted and personalized treatment strategies for individuals battling treatment-resistant depression.
The ability to visualize and quantify AMPARs could lead to the identification of individuals with specific AMPAR profiles who are predisposed to respond well to ketamine. This could also open doors to the development of novel therapeutics that specifically target AMPARs or related pathways, potentially offering even more refined and effective treatment options. The future of depression treatment may lie in such personalized approaches, moving away from a one-size-fits-all model towards therapies tailored to the individual neurobiological characteristics of each patient.
The journey from understanding basic brain function to developing effective treatments for complex psychiatric disorders is often arduous. However, this study represents a significant leap forward, offering not only a deeper understanding of an existing treatment but also a potential roadmap for future therapeutic innovation. By illuminating the intricate dance of neurotransmitters and receptors within the brain, Professor Takahashi and his team have provided a critical piece of the puzzle in the ongoing battle against treatment-resistant depression, offering renewed hope for millions worldwide.
This research was made possible through substantial funding from several key Japanese scientific and governmental organizations, including the Ministry of Education, Culture, Sports, Science and Technology (Special Coordination Funds for Promoting Science and Technology); the Japan Agency for Medical Research and Development (AMED) under grant numbers JP18dm0207023, JP19dm0207072, JP24wm0625304, JP25gm7010019, and JP20dm0107124; the Japan Society for the Promotion of Science KAKENHI with grant numbers 22H03001, 20H00549, 20H05922, 23K10432, 19H03587, 20K20603, 22K15793, and 21K07508; the Takeda Science Foundation; the Keio Next-Generation Research Project Program; the SENSHIN Medical Research Foundation; and the Japan Research Foundation for Clinical Pharmacology. The collaborative efforts and robust financial support underscore the national commitment to advancing mental health research in Japan.