Researchers at Johns Hopkins Medicine, bolstered by significant new funding from the National Institutes of Health (NIH), are making substantial strides in a groundbreaking approach to treating Alzheimer’s disease. This innovative research centers on a protein found in the brain, known for producing a minuscule yet critically important gas, that is now showing promise in safeguarding cognitive function. The study, published in the prestigious journal Proceedings of the National Academy of Sciences, delves into the intricate mechanisms by which this protein influences memory formation and its potential to combat neurodegenerative decline.
Unveiling the Role of Cystathionine β-lyase and Hydrogen Sulfide
The protein at the heart of this research is Cystathionine β-lyase, or CSE. While perhaps best known for its role in generating hydrogen sulfide – the gas notorious for its distinctive “rotten egg” odor – CSE is emerging as a key player in the complex processes of memory formation. Findings from experiments conducted on genetically engineered mice, spearheaded by study leader Bindu Paul, M.S., Ph.D., an associate professor of pharmacology, psychiatry, and neuroscience at the Johns Hopkins University School of Medicine, underscore this protein’s significance.
The overarching goal of this research endeavor is to deepen our understanding of CSE’s functionality and to ascertain whether augmenting its activity could serve as a protective mechanism for brain cells, thereby slowing the progression of devastating neurodegenerative diseases like Alzheimer’s.
Hydrogen Sulfide: A Potential Guardian of Neuronal Health
Prior scientific investigations have offered compelling evidence suggesting that hydrogen sulfide, when present at precise levels, can confer protective benefits to neurons in animal models. However, a significant hurdle has been the inherent toxicity of hydrogen sulfide in larger concentrations, rendering direct delivery to the brain unsafe. Consequently, the scientific community has shifted its focus toward deciphering methods to safely maintain the extremely low concentrations of this gas that are naturally present within neurons.
The latest findings from the Johns Hopkins team reveal a striking correlation: mice engineered to be deficient in the CSE enzyme exhibited marked impairments in memory and learning capabilities. Furthermore, these mice displayed elevated levels of oxidative stress, significant DNA damage, and compromised integrity of the blood-brain barrier. These physiological alterations are recognized as characteristic hallmarks commonly associated with Alzheimer’s disease, according to Dr. Paul, the study’s corresponding author.
A Legacy of Research Paving the Way Forward
This current body of work stands as a testament to years of dedicated research, building upon foundational discoveries made by the team of Solomon Snyder, M.D., D.Sc., D.Phil., professor emeritus of neuroscience, pharmacology, and psychiatry. In 2014, Dr. Snyder’s group published seminal findings indicating that CSE played a supportive role in brain health within mice afflicted with Huntington’s disease. Their research utilized mice genetically modified to lack the CSE protein, a strain initially developed in 2008 when the protein’s connection to cardiovascular functions, specifically blood vessel function and blood pressure regulation, was first elucidated.
Further advancing this line of inquiry, the group observed in 2021 that CSE was not functioning optimally in mice modeling Alzheimer’s disease. Crucially, they also found that very small, controlled injections of hydrogen sulfide proved beneficial in protecting brain function in these models.
While these earlier studies often focused on mice possessing additional genetic mutations predisposing them to neurodegenerative conditions, the latest research uniquely isolates and scrutinizes the specific role of CSE itself. This meticulous approach allows for a clearer understanding of CSE’s intrinsic contribution to cognitive health.
"This most recent work indicates that CSE alone is a major player in cognitive function and could provide a new avenue for treatment pathways in Alzheimer’s disease," stated Dr. Snyder, a co-corresponding author who retired from the Johns Hopkins Medicine faculty in 2023. His continued involvement underscores the enduring importance of this research area.
The Tangible Link Between Memory Deficits and CSE Deficiency
To meticulously investigate the intricate ways in which CSE influences memory, the scientists conducted a series of comparative experiments. They utilized the same genetically engineered mouse strain developed in 2008, pitting mice lacking the CSE protein against their normal counterparts. A key assessment involved evaluating spatial memory – the ability to recall directions and navigate based on environmental cues – employing a well-established behavioral paradigm known as the Barnes maze.
In this experimental setup, mice are trained to locate a hidden escape route to avoid a bright, aversive light. At two months of age, both the control mice and those deficient in CSE demonstrated comparable performance, successfully finding the shelter within a three-minute timeframe. However, a significant divergence emerged by the six-month mark. The CSE-deficient mice began to struggle considerably in locating the escape route, exhibiting a clear decline in their spatial memory, while the normal mice maintained their proficiency.
"The decline in spatial memory indicates a progressive onset of neurodegenerative disease that we can attribute to CSE loss," explained Suwarna Chakraborty, the study’s first author and a researcher in Dr. Paul’s laboratory. This observation provides a direct, albeit animal-based, parallel to the cognitive deterioration experienced by individuals with Alzheimer’s.
Cellular and Structural Brain Changes Mimicking Alzheimer’s Pathology
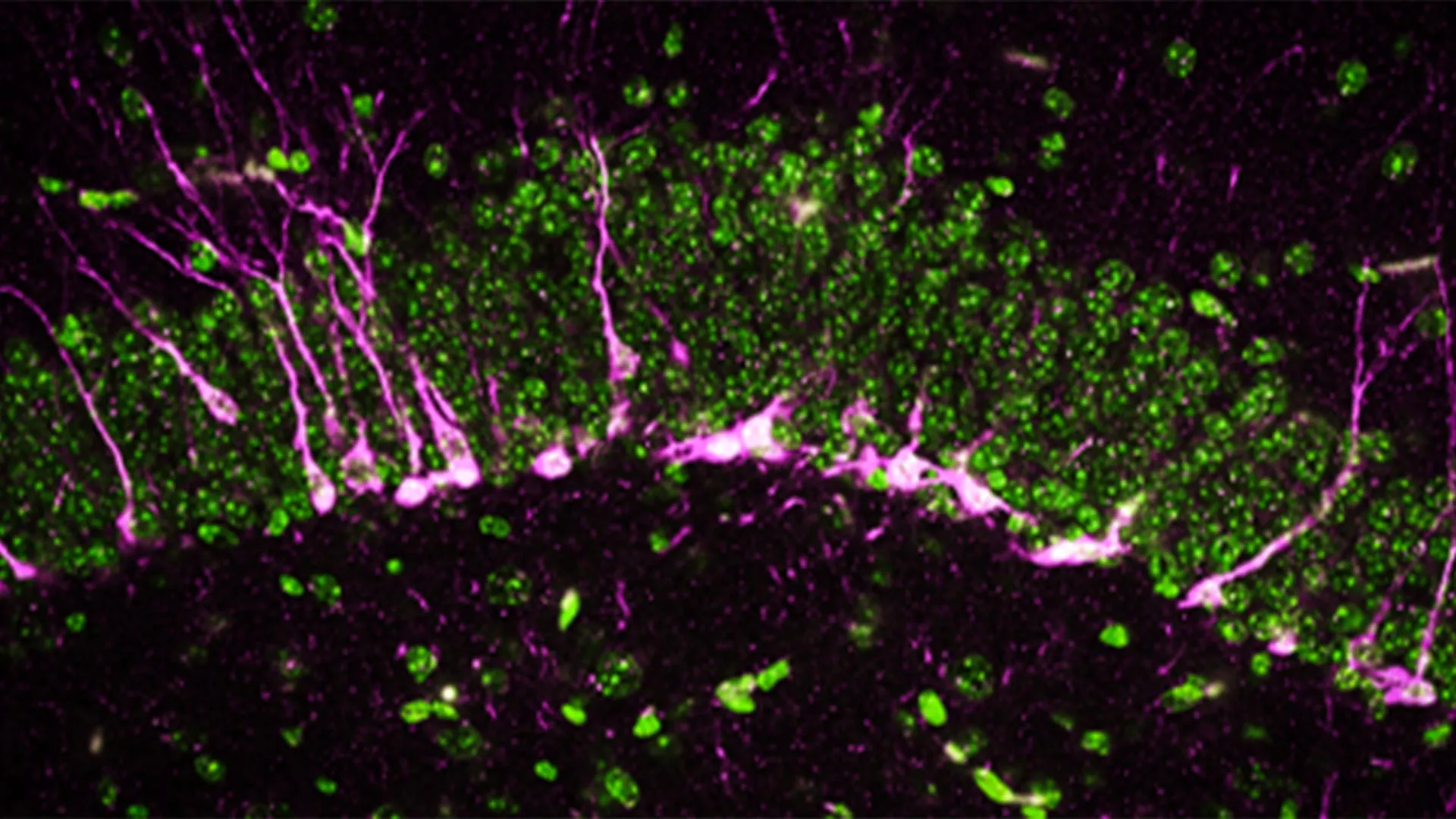
Beyond behavioral assessments, the researchers delved into the cellular and structural impacts of CSE absence on the brain. The hippocampus, a brain region indispensable for learning and memory consolidation, relies heavily on the continuous generation of new neurons, a process known as neurogenesis. Disruptions to this vital process are a well-documented characteristic of numerous neurodegenerative diseases, including Alzheimer’s.
Employing a combination of sophisticated biochemical and analytical methodologies, the team discovered that key proteins essential for neurogenesis were either reduced in quantity or entirely absent in the brains of mice lacking CSE.
Further microscopic examinations, conducted using high-powered electron microscopes, revealed striking structural damage within the brains of these CSE-deficient mice. The scientists observed substantial discontinuities and breaks in the cerebral blood vessels, a clear indication of compromised blood-brain barrier integrity – another cardinal feature of Alzheimer’s pathology. Moreover, newly formed neurons in these mice encountered significant difficulties in migrating to the hippocampus, where their normal function is to contribute to the formation of new memories.
"The mice lacking CSE were compromised at multiple levels, which correlated with symptoms that we see in Alzheimer’s disease," remarked Sunil Jamuna Tripathi, a co-first author and researcher in Dr. Paul’s lab. This multi-faceted cellular damage paints a stark picture of the profound impact of CSE deficiency on brain health.
The Broader Implications for Alzheimer’s Therapeutics
Alzheimer’s disease represents a formidable public health challenge, affecting over six million individuals in the United States alone, with projections indicating a continued upward trend, according to the U.S. Centers for Disease Control and Prevention. The current therapeutic landscape offers limited options, with no treatments consistently demonstrating the ability to halt or significantly slow the disease’s relentless progression.
The findings from Johns Hopkins Medicine offer a beacon of hope by illuminating a novel therapeutic avenue. The researchers posit that targeting CSE and its endogenous production of hydrogen sulfide could pave the way for the development of innovative therapies. Such therapies would aim to bolster brain cell resilience, protect vital cognitive functions, and ultimately decelerate the debilitating trajectory of Alzheimer’s disease. This approach moves beyond simply clearing pathological protein aggregates, a strategy that has met with mixed success, and instead focuses on bolstering the brain’s intrinsic defense mechanisms.
A Robust Foundation of Support: Funding and Collaboration
The significant progress achieved in this research is made possible by substantial financial backing from a consortium of esteemed institutions. Primary support has been provided by the National Institutes of Health (NIH) through several grants, including 1R01AG071512, P50 DA044123, 1R21AG073684, O1AGs066707, U01 AG073323, AG077396, NS101967, NS133688, and P01CA236778. Additional crucial funding has come from the Department of Defense (HT94252310443), the American Heart Association, the AHA-Allen Initiative in Brain Health and Cognitive Impairment, the Solve ME/CFS Initiative, the Catalyst Award from Johns Hopkins University, the Valour Foundation, the Wick Foundation, a Department of Veterans Affairs Merit Award (I01BX005976), the Louis Stokes Cleveland Department of Medical Affairs Veterans Center, the Mary Alice Smith Funds for Neuropsychiatry Research, the Lincoln Neurotherapeutics Research Fund, and the Gordon and Evie Safran Neuropsychiatry Fund. Funding from the Leonard Krieger Fund of the Cleveland Foundation further underscores the broad commitment to this vital research.
This multidisciplinary effort has brought together a distinguished group of scientists from various institutions. Beyond Drs. Paul, Snyder, Chakraborty, and Tripathi from Johns Hopkins, the research team includes Richa Tyagi and Benjamin Orsburn from Johns Hopkins; Edwin Vázquez-Rosa, Kalyani Chaubey, Hisashi Fujioka, Emiko Miller, and Andrew Pieper from Case Western University; Thibaut Vignane and Milos Filipovic from the Leibniz Institute for Analytical Sciences in Germany; Sudarshana Sharma from Hollings Cancer Center; Bobby Thomas from Darby Children’s Research Institute and the Medical University of South Carolina; and Zachary Weil and Randy Nelson from West Virginia University School of Medicine. This extensive collaboration highlights the complex and multi-institutional nature of cutting-edge scientific discovery.
The implications of this research extend beyond the immediate pursuit of an Alzheimer’s treatment. Understanding the fundamental role of CSE in cognitive function could have broader applications for other neurodegenerative conditions characterized by memory impairment. As the global population ages, the need for effective interventions against diseases like Alzheimer’s becomes increasingly urgent, making research like this not just scientifically significant, but profoundly vital for public health. The journey from laboratory discovery to clinical application is often long and arduous, but this latest advancement marks a critical and promising step forward.