The intricate, often overlooked sense of smell, a cornerstone of our daily experience, has long eluded complete scientific understanding. From alerting us to danger to enriching our appreciation of food and triggering profound emotional memories, olfaction plays a vital role in our lives. Despite its pervasive influence, the fundamental biological mechanisms underpinning how we detect and interpret scents have remained a significant enigma, particularly when compared to the more thoroughly mapped sensory systems of vision, hearing, and touch. This gap in knowledge has now been dramatically narrowed with the groundbreaking creation of the first detailed map of smell receptors within the mammalian nose, a discovery that challenges decades of scientific assumptions and opens new avenues for understanding and potentially treating olfactory disorders.
Decades of Mystery: The Elusive Olfactory Map
For generations, scientists have grappled with the sheer complexity of the olfactory system. While the organizational principles of sensory receptors in the eyes, ears, and skin have been elucidated, the nose has remained a persistent outlier. "Olfaction has been the one exception; it’s the sense that has been missing a map for the longest time," stated Sandeep (Robert) Datta, a professor of neurobiology at Harvard Medical School and the senior author of the pivotal new study.
The challenge stems from the system’s staggering complexity. A single mouse, for instance, possesses approximately 20 million olfactory neurons. Each of these neurons is equipped with one of over a thousand distinct types of smell receptors. This contrasts sharply with human color vision, which relies on a mere three primary receptor types. Each specific smell receptor is designed to detect a particular subset of odor molecules, rendering the olfactory system an exceptionally intricate network.
The journey to map these receptors began in earnest in 1991 with the initial identification of smell receptors. Over the subsequent decades, researchers diligently sought to uncover organizational patterns in their distribution. Early investigations suggested a rather diffuse arrangement, with receptors clustered into only a few broad zones, leading to a prevailing hypothesis that their placement within the nasal cavity was largely random. This prevailing notion of randomness presented a significant conceptual barrier, implying that the brain might have to sift through a chaotic signal to decipher specific smells.
A Quantum Leap in Resolution: Unveiling Hidden Organization
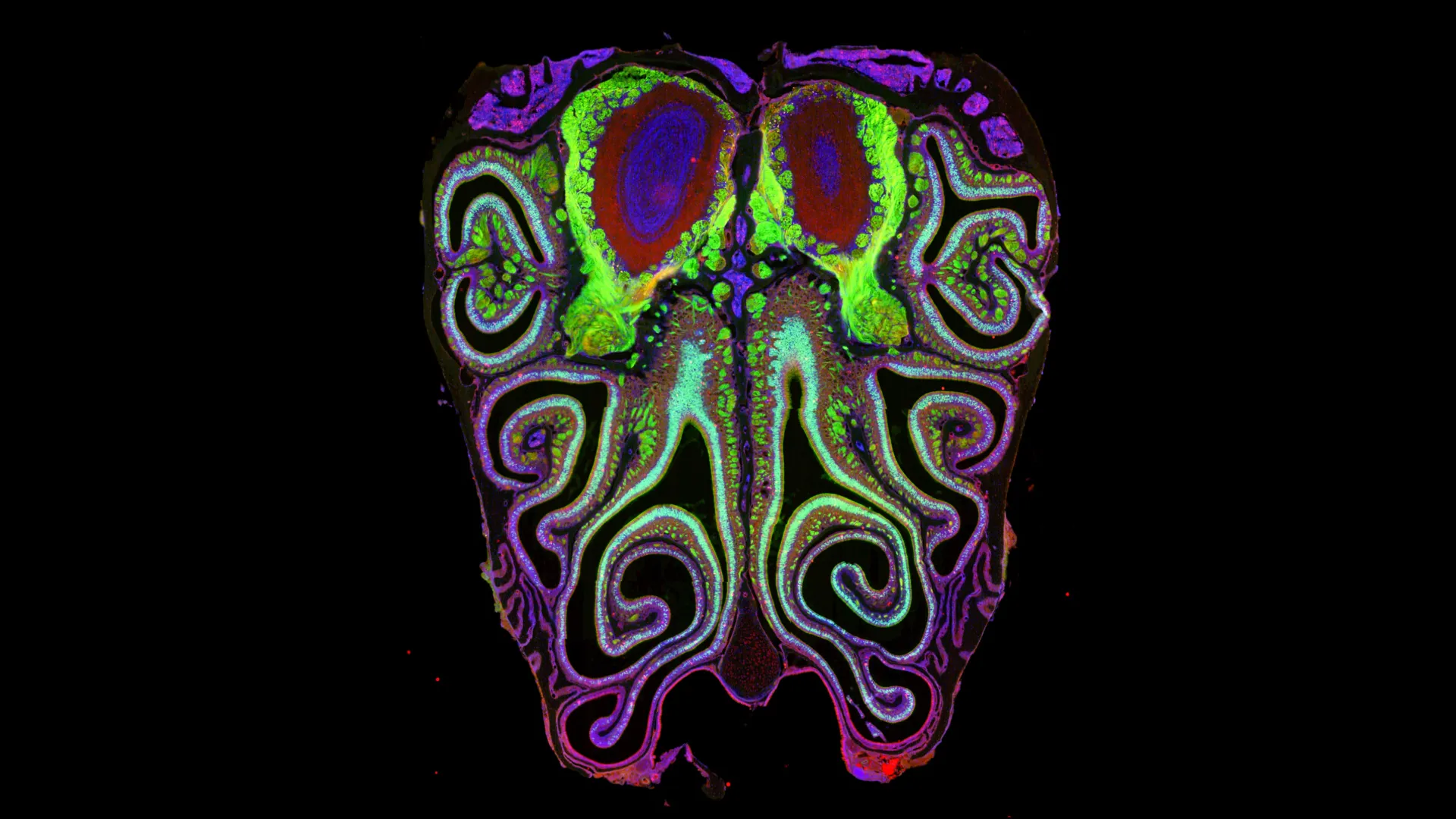
The advent of advanced genetic and imaging technologies has now enabled a far more granular examination. Datta and his team leveraged these powerful new tools to revisit the long-standing question of olfactory receptor organization. Their ambitious study, which meticulously analyzed approximately 5.5 million neurons across more than 300 mice, represents a monumental undertaking in neural tissue sequencing and spatial mapping.
The researchers employed a sophisticated combination of single-cell sequencing, a technique that precisely identifies which receptor type each individual neuron expresses, and spatial transcriptomics, which determines the exact three-dimensional location of these neurons within the nasal epithelium. "This is now arguably the most sequenced neural tissue ever, but we needed that scale of data in order to understand the system," Datta emphasized, underscoring the sheer volume of data required to discern subtle patterns within millions of cells.
The results of this extensive analysis were nothing short of revelatory. Instead of the expected randomness, the study unveiled a remarkably ordered and consistent pattern. The olfactory neurons, far from being scattered haphazardly, are organized into tightly packed, overlapping horizontal stripes. These stripes run consistently from the top to the bottom of the nasal cavity, with neurons expressing the same receptor type forming distinct bands. This highly organized arrangement was found to be virtually identical across all the mice studied, a testament to the conserved nature of this biological architecture.
Bridging the Gap: From Nose to Brain
Crucially, the researchers discovered that this intricate map within the nose is not an isolated phenomenon. Their findings demonstrate a striking alignment between the organization of receptor stripes in the nasal epithelium and the corresponding neural maps found in the olfactory bulb, the first processing center for scent information in the brain. This direct correspondence provides unprecedented insight into how odor signals are systematically transmitted from the periphery into the complex neural circuits of the brain.
"Our results bring order to a system that was previously thought to lack order, which changes conceptually how we think this works," Datta stated. This paradigm shift suggests that the brain may not need to decipher a chaotic signal but rather receives a pre-organized representation of olfactory information, simplifying the initial stages of scent perception.
The Developmental Blueprint: How the Map Takes Shape
Beyond detailing the final organizational structure, the study also delved into the developmental processes that give rise to this precise olfactory map. The research team identified retinoic acid, a crucial molecule known for its role in regulating gene activity and developmental processes, as a key orchestrator of this organization.
They found that a gradient of retinoic acid within the developing nose plays a pivotal role in guiding olfactory neurons. This molecular gradient appears to direct each neuron to activate the correct smell receptor based on its precise positional information. To validate this hypothesis, the researchers experimentally altered the levels of retinoic acid. The results were dramatic: the entire receptor map shifted, either upward or downward, demonstrating the molecule’s critical influence on establishing the striped organization. "We show that development can achieve this feat of organizing a thousand different smell receptors into an incredibly precise map that’s consistent across animals," Datta explained.
This work builds upon and corroborates findings from a parallel study also published in the prestigious journal Cell on April 28th, led by Catherine Dulac, a distinguished professor at Harvard University. The convergence of these independent research efforts lends significant weight to the discovery of this fundamental olfactory organization.
Implications for Human Health: Restoring a Vital Sense
The implications of this discovery extend far beyond the realm of basic neuroscience. The ability to perceive smell is profoundly impactful on human health, safety, and well-being. Loss of smell, known as anosmia, can have devastating consequences, affecting an individual’s ability to detect dangers like gas leaks or spoiled food, diminishing the pleasure derived from eating, and contributing to social isolation and depression. Currently, effective treatments for olfactory dysfunction are scarce.
"We cannot fix smell without understanding how it works on a basic level," Datta asserted. This newly established map provides the foundational knowledge necessary to develop targeted interventions. The research team is now focused on understanding the precise sequence and rationale behind the ordered appearance of these receptor stripes and, critically, whether this same organizational principle is conserved in humans.
The potential applications are vast. This knowledge could pave the way for novel therapeutic strategies, including the development of advanced stem cell therapies designed to regenerate damaged olfactory tissues or the creation of sophisticated brain-computer interfaces that can translate artificial olfactory signals into perceived smells. Such advancements could offer hope to millions suffering from smell loss due to aging, viral infections, neurodegenerative diseases, or head injuries.
"Smell has a really profound and pervasive effect on human health, so restoring it is not just for pleasure and safety but also for psychological well-being," Datta concluded. "Without understanding this map, we’re doomed to fail in developing new treatments." This breakthrough marks a significant turning point in our quest to fully comprehend and potentially restore one of our most fundamental sensory connections to the world.
A Collaborative Effort and Future Directions
The research leading to this groundbreaking discovery was a collaborative endeavor involving numerous scientists. The core team included David Brann, Tatsuya Tsukahara, Cyrus Tau, Dennis Kalloor, Rylin Lubash, Lakshanyaa Kannan, Nell Klimpert, Mihaly Kollo, Martin Escamilla-Del-Arenal, Bogdan Bintu, Andreas Schaefer, Alexander Fleischmann, and Thomas Bozza.
Funding for this extensive research was generously provided by grants from the National Institutes of Health (R01DC021669, R01DC021422, R01DC021965, and F31DC019017), the Yang Tan Collective at Harvard, and a National Science Foundation Graduate Research Fellowship. These contributions underscore the significant investment required for such complex and high-impact scientific inquiry.
The future directions of this research are clear: to translate this fundamental understanding into tangible benefits for human health. The ongoing investigation into the specifics of receptor stripe ordering and its presence in humans promises to unlock new therapeutic avenues and deepen our appreciation for the intricate biological symphony that allows us to perceive the world through scent.