A groundbreaking study published in The Lancet Digital Health is shedding new light on the brain’s remarkable ability to adapt following a stroke, even in cases of severe physical impairment. Researchers at the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have discovered that in individuals experiencing significant motor deficits, certain areas of the brain not directly affected by the stroke may exhibit characteristics of a "younger" structure. This phenomenon appears to be a sophisticated compensatory mechanism, reflecting the brain’s inherent capacity for reorganization and rewiring after injury.
The research, a significant undertaking by the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, analyzed a vast dataset comprising brain scans from over 500 stroke survivors. These invaluable data points were meticulously collected from 34 distinct research centers spanning eight countries, creating an unprecedented global repository of stroke recovery information. By employing advanced deep learning models, specifically trained on tens of thousands of magnetic resonance imaging (MRI) scans, the scientific team was able to estimate the "brain age" of numerous regions within each hemisphere of the brain. This sophisticated analysis allowed them to dissect how stroke impacts both the structural integrity of the brain and the subsequent recovery process.
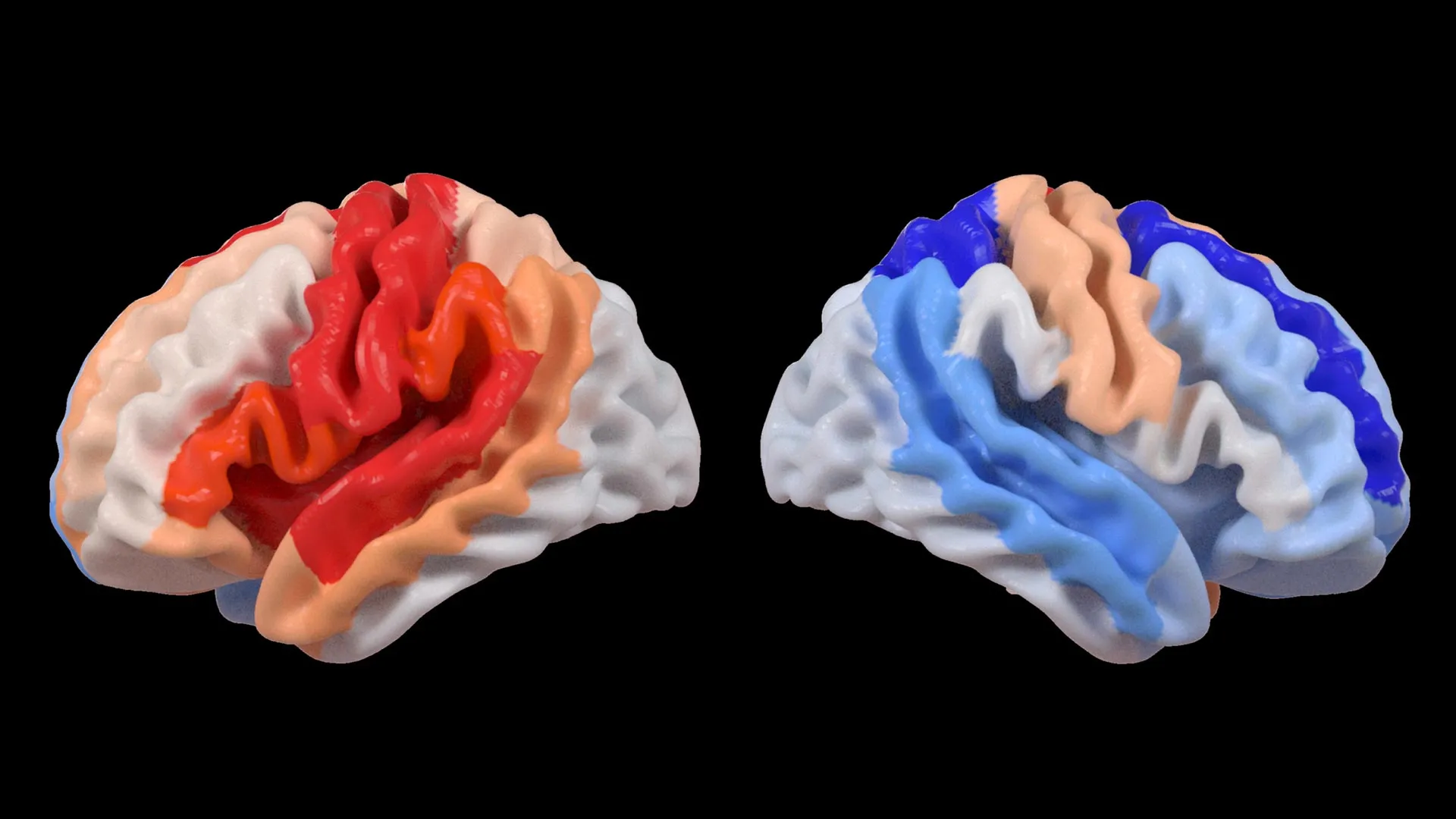
"Our findings revealed a striking duality," explained Hosung Kim, PhD, an associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author of the study. "While larger strokes demonstrably accelerate aging in the hemisphere directly affected by the damage, we observed a paradoxical rejuvenation, or a ‘younger’ appearance, in corresponding areas of the opposite, undamaged hemisphere. This distinct pattern strongly suggests that the brain is actively reorganizing itself, essentially revitalizing undamaged neural networks to compensate for the functional losses incurred by the stroke."
AI Uncovers the Brain’s Rewiring Blueprint Post-Stroke
At the heart of this discovery lies the application of a sophisticated artificial intelligence technique known as a graph convolutional network. This AI system was instrumental in estimating the biological age of 18 distinct brain regions based on detailed MRI data. The predicted age for each region was then meticulously compared with the individual’s actual chronological age. The resulting metric, termed the brain-predicted age difference (brain-PAD), serves as a crucial indicator of overall brain health and integrity.
When these brain age measurements were correlated with objective assessments of motor function, a clear and compelling pattern emerged. Stroke survivors who exhibited severe impairments in movement, even after undergoing more than six months of intensive rehabilitation, consistently displayed a younger-than-expected brain age in regions situated on the side of the brain opposite to the initial stroke lesion. This effect was particularly pronounced within the frontoparietal network, a critical brain system responsible for a complex array of functions including movement planning, attentional control, and coordinated action.
"These findings provide compelling evidence that when stroke-induced damage leads to significant loss of motor control, undamaged regions on the opposite side of the brain appear to adapt and actively contribute to compensating for these deficits," Dr. Kim elaborated. "We observed this phenomenon most clearly in the contralesional frontoparietal network, which exhibited a more ‘youthful’ structural pattern. This network is well-established for its role in supporting motor planning, attention, and coordination. Rather than directly indicating a full restoration of motor function, this observed pattern likely reflects the brain’s sophisticated attempt to adjust and maintain essential operations when the primary damaged motor system is no longer capable of functioning normally. This represents a novel perspective on neuroplasticity, revealing adaptive changes that were previously undetectable with traditional imaging techniques."
A Global Collaborative Effort Magnifies Subtle Neural Patterns
The profound insights derived from this study are intrinsically linked to the robust foundation provided by the ENIGMA initiative. ENIGMA is a globally recognized collaborative network that consolidates neuroimaging data from over 50 countries, aiming to foster a deeper and more comprehensive understanding of the human brain across a spectrum of neurological conditions. By rigorously standardizing MRI data and clinical information from a multitude of research groups, the team successfully constructed the most extensive stroke neuroimaging dataset of its kind ever assembled.
"The power of pooling data from hundreds of stroke survivors across the globe and applying cutting-edge artificial intelligence is immense," stated Arthur W. Toga, PhD, director of the Stevens INI and Provost Professor at USC. "This approach enables us to detect subtle, yet significant, patterns of brain reorganization that would undoubtedly remain invisible within smaller, more localized studies. These findings, detailing regionally differential brain aging in chronic stroke patients, hold the potential to revolutionize personalized rehabilitation strategies in the future."
The timeline of this research highlights the collaborative and iterative nature of scientific advancement. The initial conceptualization of the ENIGMA Stroke Recovery Working Group likely began several years prior, with the arduous process of data acquisition, standardization, and ethical approvals taking place over an extended period. The recruitment of over 500 stroke survivors across 34 research centers is a testament to the dedication of researchers and participants worldwide. The deep learning models themselves would have undergone extensive development and validation before being applied to the collected dataset. The final analysis and interpretation of the results, culminating in the publication in The Lancet Digital Health, represent the culmination of a multi-year scientific endeavor.
Charting a Course Towards Personalized Stroke Rehabilitation
Looking ahead, the researchers are committed to extending their work by embarking on longitudinal studies. This next phase will involve meticulously tracking patients over time, from the acute stages immediately following a stroke through their long-term recovery trajectories. By continuously monitoring how brain aging patterns and structural changes evolve, clinicians will be better equipped to tailor rehabilitation treatments to the unique recovery process of each individual. The ultimate goal of this personalized approach is to significantly improve patient outcomes and enhance their overall quality of life.
The implications of this research are far-reaching. For individuals recovering from stroke, understanding these adaptive brain mechanisms offers a new perspective on the resilience of the human nervous system. For clinicians and therapists, these findings could lead to the development of more targeted and effective rehabilitation protocols. By identifying specific brain regions exhibiting compensatory "youthful" characteristics, therapists might be able to design interventions that further stimulate and leverage these undamaged networks, optimizing the recovery of lost motor functions.
The study’s methodology, leveraging AI to analyze brain age, represents a significant leap forward in neuroimaging. Traditional methods often focus on gross structural changes or lesion volume. However, this new approach delves into more nuanced indicators of neural health and adaptability, revealing functional reorganizations that might otherwise go unnoticed. This advanced analytical capability opens doors for earlier and more precise identification of recovery potential and challenges.
The broader context of stroke research underscores the critical need for such innovative studies. Stroke remains a leading cause of long-term disability globally, impacting millions of lives each year. The economic and social burden of stroke is substantial, making advancements in recovery and rehabilitation a public health imperative. The ENIGMA initiative’s success in aggregating global data is a powerful example of how international collaboration can accelerate scientific discovery in addressing major health challenges.
The funding for this pivotal research, primarily from the National Institutes of Health (NIH) grant R01 NS115845, highlights the sustained commitment of governmental bodies to advancing our understanding of neurological disorders. The support from international collaborators at institutions such as the University of British Columbia, Monash University, Emory University, and the University of Oslo underscores the truly global nature of this scientific endeavor. Their collective expertise and data contributions were indispensable in achieving the scale and depth of this study.
The potential for personalized medicine in stroke recovery is immense. By understanding the unique patterns of brain adaptation in each patient, therapists can move away from a one-size-fits-all approach. This could involve utilizing specific forms of physical therapy, neurofeedback techniques, or even pharmacological interventions that are optimized for an individual’s neurobiological profile. The "brain age" metric, as developed in this study, could become a valuable tool in a clinician’s arsenal, providing objective insights into the brain’s current state and its capacity for further adaptation.
Future research may also explore whether interventions aimed at promoting brain health and plasticity can influence these observed "younger" brain age patterns in the contralesional hemisphere. Could lifestyle factors, specific exercise regimens, or novel therapeutic approaches actively encourage this compensatory rejuvenation? These are critical questions that the ongoing work by the Stevens INI and its collaborators aims to address.
The availability of a video created by the Stevens INI, explaining the associations between contralesional neuroplasticity and motor impairment, further democratizes access to this complex scientific information. This initiative to communicate research findings through accessible formats is crucial for public engagement and for fostering a greater understanding of the brain’s remarkable capabilities.
In summary, the study published in The Lancet Digital Health represents a significant advancement in our understanding of stroke recovery. By harnessing the power of artificial intelligence and global data collaboration, researchers have uncovered a compelling phenomenon of contralesional brain rejuvenation, offering new hope and a promising path towards more personalized and effective rehabilitation strategies for stroke survivors worldwide. The continuous exploration of these adaptive mechanisms promises to unlock new avenues for improving functional outcomes and restoring quality of life for those affected by this devastating condition.