A groundbreaking study published in The Lancet Digital Health is reshaping our understanding of the brain’s remarkable capacity to adapt following a stroke. Researchers at the USC Mark and Mary Stevens Neuroimaging and Informatics Institute (Stevens INI) have uncovered evidence that individuals experiencing significant physical impairments after a stroke may exhibit structural characteristics of a "younger" brain in areas that have remained untouched by the injury. This phenomenon appears to be a testament to the brain’s inherent ability to reorganize and compensate for lost function, a process known as neuroplasticity.
Unraveling the Brain’s Adaptive Mechanisms
The research, a significant undertaking by the Enhancing NeuroImaging Genetics through Meta-Analysis (ENIGMA) Stroke Recovery Working Group, involved a comprehensive analysis of brain scans from over 500 stroke survivors. These invaluable data were meticulously collected from 34 research centers spanning eight countries, creating an unprecedented global dataset. By leveraging sophisticated deep learning models, trained on tens of thousands of magnetic resonance imaging (MRI) scans, the scientific team was able to estimate the "brain age" of distinct regions within each hemisphere of the brain. This innovative approach allowed them to meticulously examine how a stroke impacts both the structural integrity of the brain and the subsequent recovery process.
"Our findings were quite astonishing," stated Hosung Kim, PhD, an associate professor of research neurology at the Keck School of Medicine of USC and a co-senior author of the study. "We observed that while larger strokes tend to accelerate the aging process in the damaged hemisphere, the opposite side of the brain, paradoxically, appears younger. This distinct pattern strongly suggests that the brain is actively reorganizing itself, essentially rejuvenating undamaged neural networks to compensate for the functional deficits caused by the stroke."
AI as a Window into Brain Rewiring
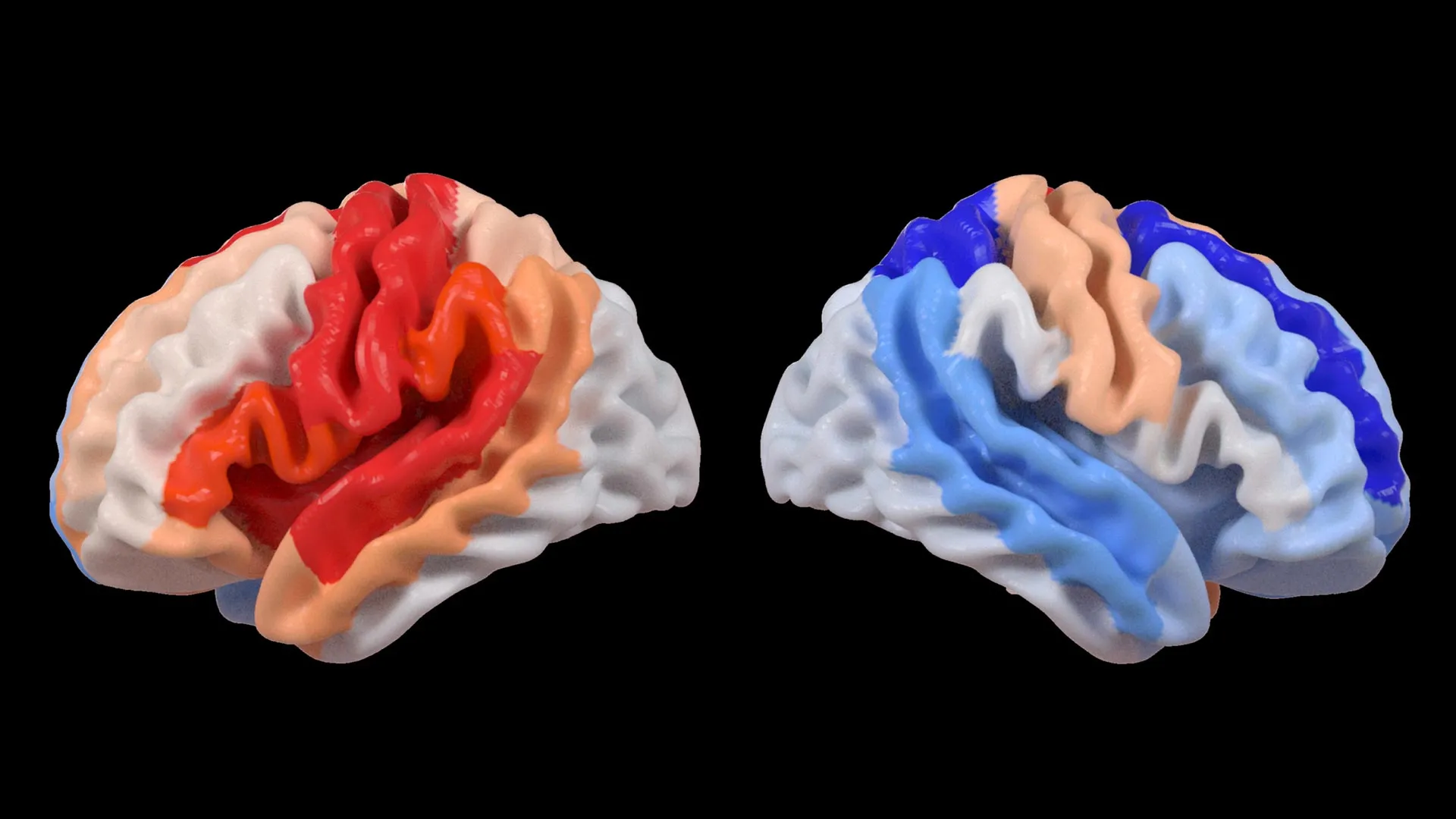
At the heart of this discovery lies a powerful artificial intelligence system: a graph convolutional network. This advanced AI was instrumental in estimating the biological age of 18 specific brain regions, drawing insights from the detailed MRI data. By comparing these AI-derived age predictions with each individual’s chronological age, researchers calculated a measure known as the brain-predicted age difference (brain-PAD). This metric serves as a crucial indicator of overall brain health and the extent of deviations from typical aging patterns.
When these brain age measurements were correlated with scores measuring motor function, a compelling and consistent pattern emerged. Stroke survivors who experienced severe impairments in their movement capabilities, even after more than six months of intensive rehabilitation, displayed a brain age in regions opposite the site of their stroke that was younger than expected. This effect was particularly pronounced in the frontoparietal network, a critical brain circuit involved in a wide array of cognitive functions including movement planning, attention, and the coordination of complex actions.
Dr. Kim elaborated on the implications of this observation: "These findings suggest a profound adaptive response within the brain. When stroke damage leads to a significant loss of motor control, undamaged regions on the contralateral side of the brain appear to step in and attempt to compensate. We specifically noted this in the contralesional frontoparietal network, which exhibited a more ‘youthful’ pattern. This network is well-known for its role in supporting motor planning, attention, and coordination. Rather than being a direct indicator of full motor recovery, this pattern likely reflects the brain’s tenacious effort to adjust and maintain function when the primary motor system is compromised and can no longer operate normally. This provides us with an entirely new lens through which to observe neuroplasticity, a level of detail that traditional imaging techniques simply could not capture."
The Power of Large-Scale Data in Uncovering Hidden Patterns
The success of this pioneering study is intrinsically linked to the ENIGMA initiative, a vast global collaboration that unites data from over 50 countries. This collective effort aims to foster a deeper and more nuanced understanding of the brain across a spectrum of neurological conditions. By meticulously standardizing MRI data and clinical information from a multitude of research groups, the team was able to construct what is arguably the most extensive stroke neuroimaging dataset of its kind ever assembled.
"By pooling data from hundreds of stroke survivors across the globe and applying cutting-edge artificial intelligence, we are able to detect incredibly subtle patterns of brain reorganization that would undoubtedly remain invisible in smaller, more localized studies," emphasized Arthur W. Toga, PhD, director of the Stevens INI and a Provost Professor at USC. "These findings, revealing regionally differential brain aging in chronic stroke patients, hold immense promise for the future development of highly personalized rehabilitation strategies."
Charting a Course Towards Personalized Stroke Recovery
The research team is not resting on their laurels. Their future endeavors are focused on longitudinal studies, where they will meticulously track patients over extended periods. This will involve monitoring their progress from the immediate aftermath of a stroke through the complex and often lengthy process of long-term recovery. By observing how these patterns of brain aging and structural changes evolve over time, clinicians may be better equipped to tailor therapeutic interventions to the unique recovery trajectory of each individual patient. The ultimate goal is to significantly improve patient outcomes and enhance their overall quality of life.
The study, titled "Deep learning prediction of MRI-based regional brain age reveals contralesional neuroplasticity associated with severe motor impairment in chronic stroke: A worldwide ENIGMA study," received substantial funding from the National Institutes of Health (NIH) under grant R01 NS115845. Furthermore, the research benefited from the invaluable contributions and collaborations of international partners from prestigious institutions including the University of British Columbia, Monash University, Emory University, and the University of Oslo, underscoring the truly global nature of this scientific advancement.
Understanding the Stroke Recovery Landscape: A Broader Context
Stroke remains a leading cause of long-term disability worldwide, affecting millions of individuals annually. The immediate aftermath of a stroke is often characterized by a sudden and devastating loss of neurological function, with physical impairments being among the most visible and debilitating. Traditional approaches to stroke rehabilitation have largely focused on retraining damaged brain areas and promoting compensatory strategies. However, the plasticity of the brain, its ability to rewire itself, has always been a key area of interest and a target for therapeutic interventions.
The advent of advanced neuroimaging techniques, such as MRI, has provided unprecedented insights into the structural and functional changes that occur in the brain after a stroke. These technologies allow researchers to visualize areas of damage, monitor recovery processes, and investigate the underlying neural mechanisms of adaptation. However, quantifying the subtle nuances of these changes, particularly in chronic stroke patients where the acute phase has long passed, has been a significant challenge.
The ENIGMA consortium has been at the forefront of addressing these challenges by aggregating and harmonizing large-scale neuroimaging datasets. This approach allows for the detection of subtle effects that might be missed in smaller studies due to variations in imaging protocols, patient populations, and analytical methods. By creating standardized datasets, ENIGMA facilitates more robust and generalizable findings, accelerating the pace of discovery in neuroscience.
The concept of "brain age" as a biomarker of brain health has gained traction in recent years. It posits that deviations from an expected age-related pattern in brain structure or function can be indicative of underlying pathology or accelerated aging. In the context of stroke, understanding how the injury impacts brain age, both in damaged and undamaged regions, provides a novel metric for assessing the severity of the insult and the brain’s adaptive response.
The frontoparietal network, highlighted in this study, is a crucial component of the brain’s executive control system. It is involved in higher-order cognitive functions such as planning, decision-making, working memory, and attention. Its significant role in motor control, particularly in the planning and execution of voluntary movements, makes it a prime candidate for involvement in compensatory mechanisms following stroke. The observation that this network shows a "younger" brain age in individuals with severe motor impairments suggests that the brain is dynamically reallocating resources and potentially enhancing the functionality of these undamaged circuits to mitigate the impact of the motor deficit.
Implications for Future Rehabilitation Strategies
The findings of this study have profound implications for the future of stroke rehabilitation. The ability to identify specific patterns of brain reorganization, such as the "younger" brain age in the contralesional frontoparietal network, could pave the way for more personalized and targeted interventions.
Personalized Treatment Plans: Instead of a one-size-fits-all approach, rehabilitation programs could be tailored based on an individual’s unique pattern of brain adaptation. For example, patients exhibiting this specific pattern might benefit from interventions that further enhance the plasticity of the frontoparietal network, focusing on motor planning and coordination exercises.
Predictive Biomarkers: The brain-PAD metric, derived from deep learning models, could potentially serve as a predictive biomarker. It might help clinicians anticipate a patient’s recovery trajectory and identify those at higher risk of persistent severe impairments, allowing for earlier and more intensive therapeutic interventions.
Optimizing Rehabilitation Timing: Understanding how brain aging patterns evolve over time post-stroke could inform the optimal timing and intensity of rehabilitation. Interventions might be most effective when aligned with specific periods of heightened neuroplasticity.
Development of Novel Therapies: This research could stimulate the development of new therapeutic strategies aimed at enhancing contralesional neuroplasticity. This might include targeted neuromodulation techniques, advanced neurofeedback protocols, or novel pharmacological interventions designed to promote brain adaptation.
The Road Ahead: Continued Research and Clinical Translation
The journey from groundbreaking research to widespread clinical application is often long and complex. However, the findings from the USC Stevens INI and the ENIGMA consortium represent a significant leap forward in our understanding of stroke recovery. The continued commitment to longitudinal studies and the translation of these AI-driven insights into clinical practice hold immense promise for improving the lives of stroke survivors worldwide. The integration of sophisticated computational tools with large-scale, multi-center datasets is proving to be an invaluable strategy for unraveling the mysteries of the human brain and developing more effective interventions for neurological disorders. The study serves as a powerful testament to the collaborative spirit of scientific inquiry and the transformative potential of artificial intelligence in medical research.