The widespread reliance on "hair porosity" as a foundational concept in haircare, particularly through popular at-home diagnostic tests, has been challenged by recent scientific insights. While the term "porosity" is a complex one with varied interpretations, many of the commonly held beliefs and diagnostic methods associated with it, such as the widely practiced float test and drop test, fundamentally misinterpret the underlying scientific principles. Instead of measuring how much water hair can absorb internally, these tests primarily reveal how water interacts with the hair’s surface due to surface tension. This clarification, articulated by beauty science communicator Dr. Michelle Wong of Lab Muffin Beauty Science, underscores the need for a more nuanced, evidence-based approach to understanding hair health and selecting appropriate haircare products.
The Pervasive Concept of Hair Porosity
In the beauty industry, hair porosity is generally defined by the hair cuticle’s ability to absorb and retain moisture. It’s often categorized into low, medium, and high porosity, with specific product recommendations linked to each category. Low porosity hair, for instance, is thought to have tightly bound cuticles that resist moisture penetration, requiring lighter products. High porosity hair, conversely, is believed to have open, damaged cuticles that readily absorb moisture but struggle to retain it, thus benefiting from heavier, more sealing products. This framework has become a cornerstone of personalized haircare advice, guiding countless consumers in their product choices. However, the scientific basis for the at-home tests used to determine these categories has long been questioned by cosmetic chemists and material scientists. The disconnect between popular belief and scientific reality highlights a significant area for educational outreach within the beauty community.

Debunking the "Waterproof Hair" Myth
A central misconception fueling the porosity myth is the idea that undamaged, healthy hair is inherently waterproof or water-resistant. This belief suggests that a pristine cuticle layer acts as an impermeable barrier, sealing out water. Conditioners are often touted as mimicking this sealing function for damaged hair. However, scientific evidence contradicts this notion. Human hair, even in its most undamaged and conditioned state, is far from waterproof. Research indicates that undamaged hair can absorb a substantial amount of water—up to 30% of its own weight—within minutes. This absorption capacity fluctuates rapidly with environmental humidity, demonstrating hair’s inherent ability to interact with and take up water from its surroundings.
For example, data compiled by Robbins in "Chemical and Physical Behavior of Human Hair" (2012) illustrates this relationship:
- At 0% relative humidity, hair absorbs 0% water.
- At 8% relative humidity, hair absorbs 3.9% water.
- At 40% relative humidity, hair absorbs 10.2% water.
- At 63% relative humidity, hair absorbs 14.8% water.
- At 86% relative humidity, hair absorbs 22.6% water.
- At 100% relative humidity, hair absorbs 31.2% water.
This significant capacity for water absorption is attributed to the structural nature of hair. The hair cuticle, often likened to overlapping shingles on a roof or scales on a pinecone, is not a continuous, sealed surface. It possesses an F-layer, a natural conditioning layer, but this layer resides primarily on the surface of each individual cuticle scale. Crucially, there are numerous microscopic gaps and interstices between these scales, providing ample pathways for water molecules to penetrate the hair shaft.
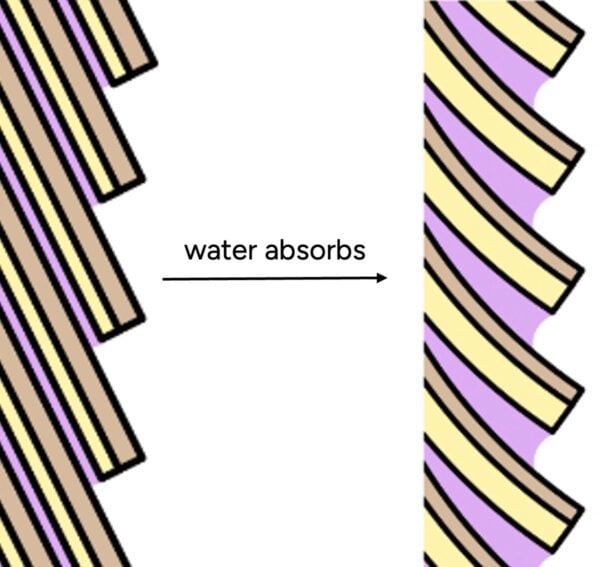
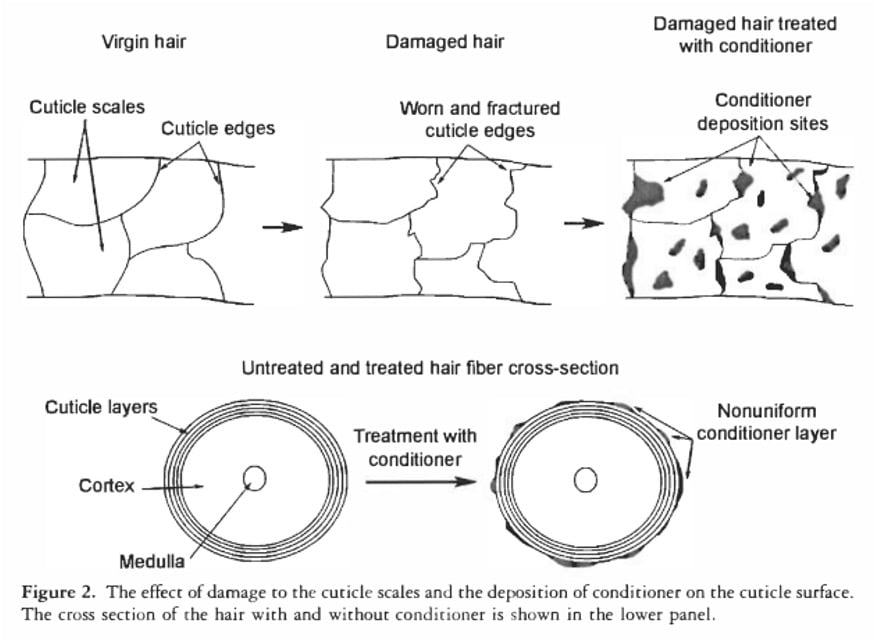
Furthermore, the role of conditioners in "sealing" the hair is also often misunderstood. Studies, such as that by La Torre and Bhushan (2006) on nanotribological effects of silicones, demonstrate that conditioners deposit onto the hair shaft not as a continuous, impenetrable film, but rather in discrete "blobs" or patches. While these deposited materials effectively smooth the hair’s surface, reduce friction, and enhance tactile softness, their discontinuous nature means they are largely ineffective at preventing the entry of tiny water molecules. The primary benefit of conditioners lies in managing the hair’s surface properties and feel, not in creating a waterproof barrier.
The Role of Surface Tension: Unpacking the At-Home Tests
Given that even healthy hair readily absorbs water, the observations from popular at-home porosity tests require an alternative explanation. This explanation lies in the principle of surface tension, which governs how liquids interact with surfaces.
Surface tension is a phenomenon where the surface of a liquid, such as water, behaves like an elastic skin. This occurs because water molecules are strongly attracted to each other through hydrogen bonds. Molecules within the bulk of the liquid are surrounded by other water molecules, forming bonds in all directions. However, molecules at the surface lack neighbours above them, leading to stronger cohesive forces with adjacent molecules along the surface and with those just below. This imbalance of forces creates a net inward pull, minimizing the surface area and resulting in a taut "skin" effect capable of supporting objects denser than water, like insects or paperclips, as long as the surface tension is not broken.
The Float Test Re-evaluated
The float test involves placing a strand of hair in a glass of water and observing whether it floats or sinks. The conventional interpretation is that hair that sinks is "high porosity" because it has quickly absorbed water, while hair that floats is "low porosity" due to its resistance to water absorption.
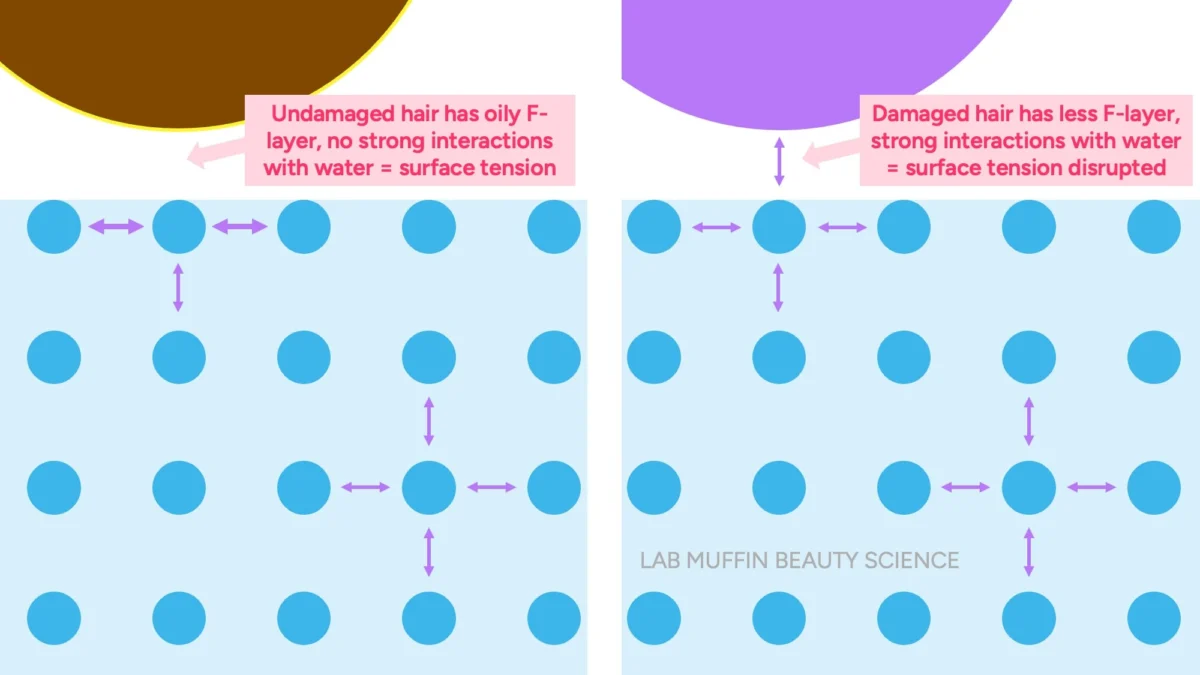
However, the scientific reality is that both hair and paperclips are denser than water and should sink. They only float if the water’s surface tension is sufficient to support them. Undamaged hair possesses an intact F-layer, an oily, hydrophobic (water-repelling) coating on its outermost cuticle scales. When this hydrophobic surface encounters water, it minimizes interaction with the water molecules, allowing the water’s surface tension to remain largely undisturbed, thus supporting the hair strand.
Conversely, damaged hair has a compromised F-layer, exposing a more hydrophilic (water-attracting) surface. When this surface comes into contact with water, it readily forms hydrogen bonds with the water molecules, effectively disrupting the cohesive forces that maintain the water’s surface tension. Once the surface tension is broken, the hair, being denser than water, will sink. Therefore, the float test primarily indicates the hydrophobicity or hydrophilicity of the hair’s surface, which is directly correlated with the level of surface damage, rather than the internal porosity or water absorption rate.

The Drop Test Re-evaluated
Similarly, the drop test, where a droplet of water is placed on a lock of hair, is also influenced by surface tension. The common observation is that on "low porosity" hair, the water forms a distinct bead, while on "high porosity" hair, it flattens out and spreads. This is typically attributed to water being unable to penetrate low porosity hair and quickly absorbing into high porosity hair.
In reality, the beading of water on undamaged hair is a classic demonstration of a hydrophobic surface repelling water, causing it to maintain a high contact angle and spherical shape due to its own strong cohesive forces (surface tension). The water does not absorb significantly into the hair shaft through this direct liquid contact in the short timeframe of the test.
When water flattens and spreads on damaged hair, it’s because the exposed, hydrophilic surface of the hair attracts the water molecules, allowing them to spread out and wet the surface more effectively. This observation is not due to rapid internal absorption of liquid water, but rather the affinity of the water for the damaged hair’s surface. While damaged hair can absorb slightly more water than undamaged hair (e.g., around 45% of its weight compared to 30%), this difference is primarily related to its internal structure and occurs over time, not instantaneously during a drop test.

Crucially, the 30% water absorption by undamaged hair (as per Robbins’ data) primarily occurs from water in its gaseous state—humidity in the air. Individual water molecules in vapor form are small enough to wiggle between the cuticle scales and enter the hair shaft without being significantly hindered by the hair’s surface chemistry or the macroscopic phenomenon of liquid surface tension. This gaseous absorption is a continuous process that governs hair’s equilibrium moisture content in different environments.
Implications for Haircare and Professional Practice
The debunking of these traditional porosity tests has significant implications for both consumers and haircare professionals. The good news is that much of the "porosity" advice that has emerged from these tests often does lead people to products that suit their hair better, but this is largely a beneficial coincidence. The tests are effectively measuring surface damage: more surface damage correlates with what is inaccurately termed "high porosity," while less surface damage is categorized as "low porosity." Haircare strategies based on addressing surface damage (e.g., using heavier conditioners, protein treatments for damaged hair; lighter products for less damaged hair) are scientifically sound and beneficial, regardless of the mislabeling.
However, the misinterpretation of these tests can lead to flawed conclusions and suboptimal haircare decisions. For instance, a common misconception is that "high porosity" hair, due to its supposed "open cuticles," will absorb chemical treatments (like dyes, perms, or relaxers) much faster. Hairdressers who rely on these porosity tests to determine processing times for chemical treatments may face issues. While surface damage does influence how chemicals initially interact with hair, the internal penetration rate of chemical agents is a distinct process that isn’t accurately predicted by surface-level observations alone. The safest and most reliable method for professionals remains conducting a strand test directly with the chemical product to gauge its efficacy and processing time on an individual’s hair.
Furthermore, external factors can skew test results. The presence of product build-up, especially from oils or certain styling agents, can create a hydrophobic layer on the hair’s surface. This can lead to a "low porosity" result in the float or drop test, even if the underlying hair structure is damaged and would otherwise be considered "high porosity." Such misleading results could direct consumers towards products that are not truly beneficial for their hair’s actual internal condition.
In conclusion, while the concept of hair porosity remains relevant in a broader scientific context (referring to the internal structure and void volume of the hair fiber), the popular at-home tests are not reliable indicators of this property. Instead, they primarily serve as proxies for the degree of surface damage and the hydrophobicity of the hair’s outermost layer. As scientific understanding advances, beauty communication, championed by experts like Dr. Michelle Wong, increasingly advocates for moving beyond simplistic and misleading tests towards a more comprehensive understanding of hair biology. Consumers are encouraged to focus on the tangible characteristics of their hair—such as dryness, oiliness, damage, and texture—when selecting products, rather than relying on tests that misinterpret fundamental scientific principles. For professionals, this clarity reinforces the importance of direct observation and empirical testing, particularly for chemical services, ensuring optimal and safe outcomes for clients.
References
Robbins CR. Chemical and Physical Behavior of Human Hair. 5th ed. Springer Berlin Heidelberg 2012.

La Torre C, Bhushan B. Nanotribological effects of silicone type, silicone deposition level, and surfactant type on human hair using atomic force microscopy. J Cosmet Sci. 2006;57(1):37-56.