The discourse surrounding hair hydration is fraught with misconceptions, often fueled by marketing rhetoric and entrenched beliefs within the beauty industry. While a plethora of products promise "hydrated hair" for a soft, smooth, and healthy feel, a deeper scientific understanding reveals a more nuanced reality, challenging both popular wisdom and some expert opinions. The prevailing notion that water is unequivocally beneficial, or conversely, inherently damaging, misses the intricate molecular dance between hair and H2O. Deciphering this interaction is crucial for effective haircare, moving beyond simplistic narratives to embrace evidence-based practices.
The Pervasive Myth of "Hydrated Hair"
The concept of "hydrated hair" as intrinsically soft, smooth, and healthy is a cornerstone of modern haircare marketing. Consumers are routinely presented with products designed to "quench" dry strands, implying that adding water directly leads to desirable textural improvements. However, scientific consensus among cosmetic chemists often diverges from this commercial narrative. Many industry professionals acknowledge the difficulty in overturning deeply ingrained consumer beliefs, opting instead to align marketing language with existing perceptions rather than embark on a costly and complex re-education effort. This strategic approach, while commercially pragmatic, perpetuates a significant scientific misunderstanding.
In reality, the sensation commonly associated with "hydrated" hair – a feeling of smoothness and softness – is often correlated with lower rather than higher water content. A pivotal study referenced in Cosmetics & Toiletries journal highlighted this paradox, where participants consistently rated hair with less intrinsic water as feeling more moisturized, smoother, less tangled, and less damaged. This counterintuitive finding underscores the disconnect between subjective sensory experience and objective scientific measurement of hair’s water content. The perception of "hydration" is, therefore, largely a tactile illusion, influenced by the hair’s surface properties rather than its internal moisture levels.
Unpacking Hair’s Molecular Architecture and Water Interaction
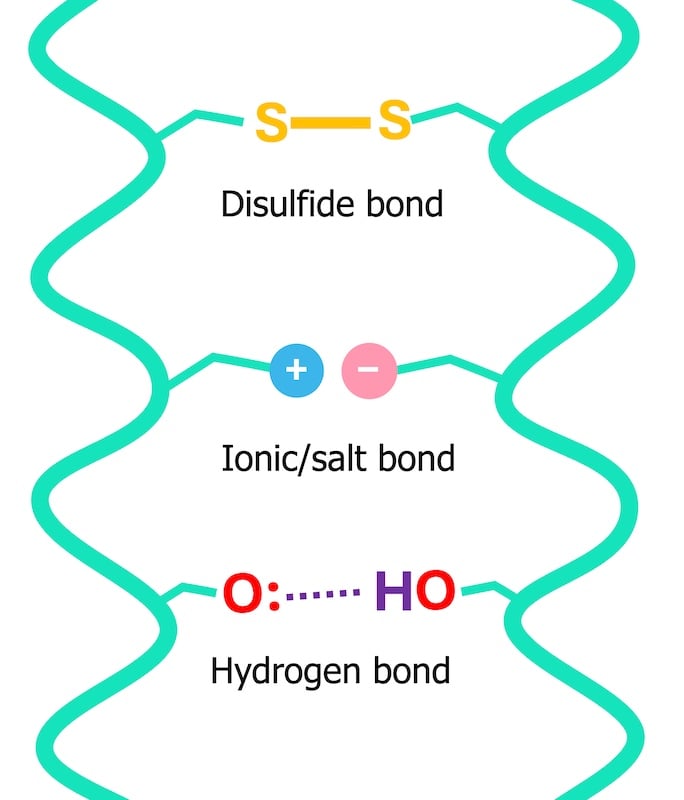
To truly grasp how water interacts with hair, one must understand its fundamental structure. Hair is primarily composed of keratin proteins, which are organized into complex fibrous structures, held together by various chemical bonds. These bonds are responsible for hair’s strength, elasticity, and overall shape. They can be broadly categorized into two types:
- Permanent Covalent Bonds: Primarily disulfide bonds, these are strong chemical links between sulfur atoms in cysteine amino acids. They are largely responsible for the permanent shape of hair and are broken or reformed during chemical treatments like perms or relaxers.
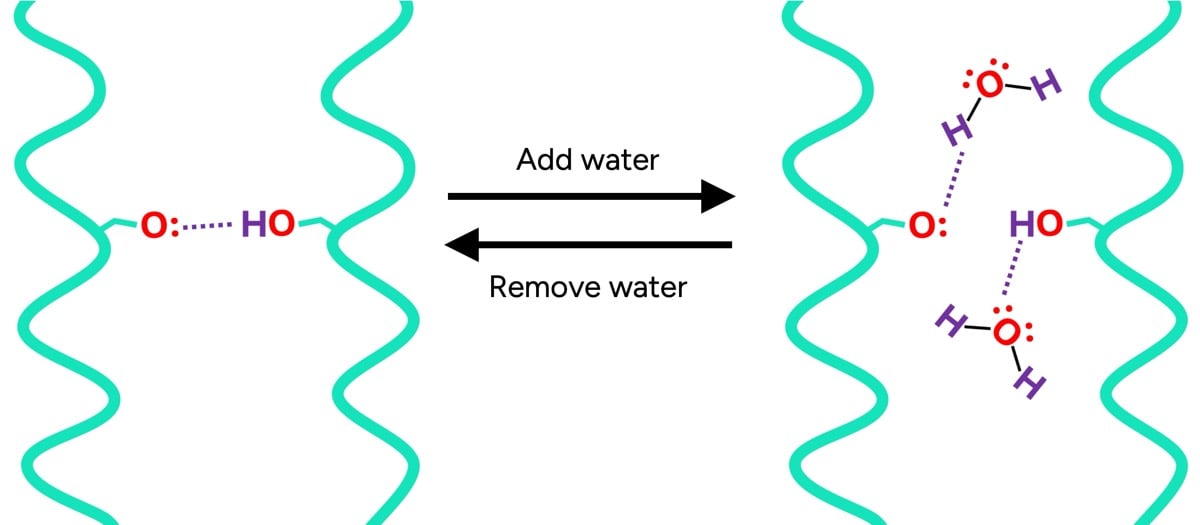
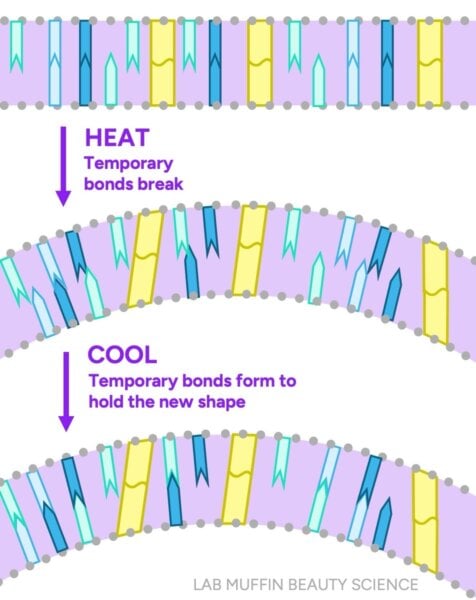
- Temporary Bonds: These include hydrogen bonds and ionic (salt) bonds. These are weaker, non-covalent interactions that form between adjacent protein chains. They are highly sensitive to water and heat, playing a crucial role in temporary styling and hair’s immediate physical properties.
The internal structure of the hair shaft, particularly the cortex, is highly hygroscopic, meaning it readily absorbs water. In dry hair, numerous temporary hydrogen bonds form between the keratin proteins, acting like molecular "handshakes" that lend stiffness and integrity. However, when water molecules are introduced, they compete for these bonding sites. Water, being a highly polar molecule, can form hydrogen bonds with the keratin proteins. This phenomenon, known as "plasticization," effectively displaces the protein-protein bonds, leading to a significant reduction in the overall number of temporary bonds holding the hair’s internal structure together.
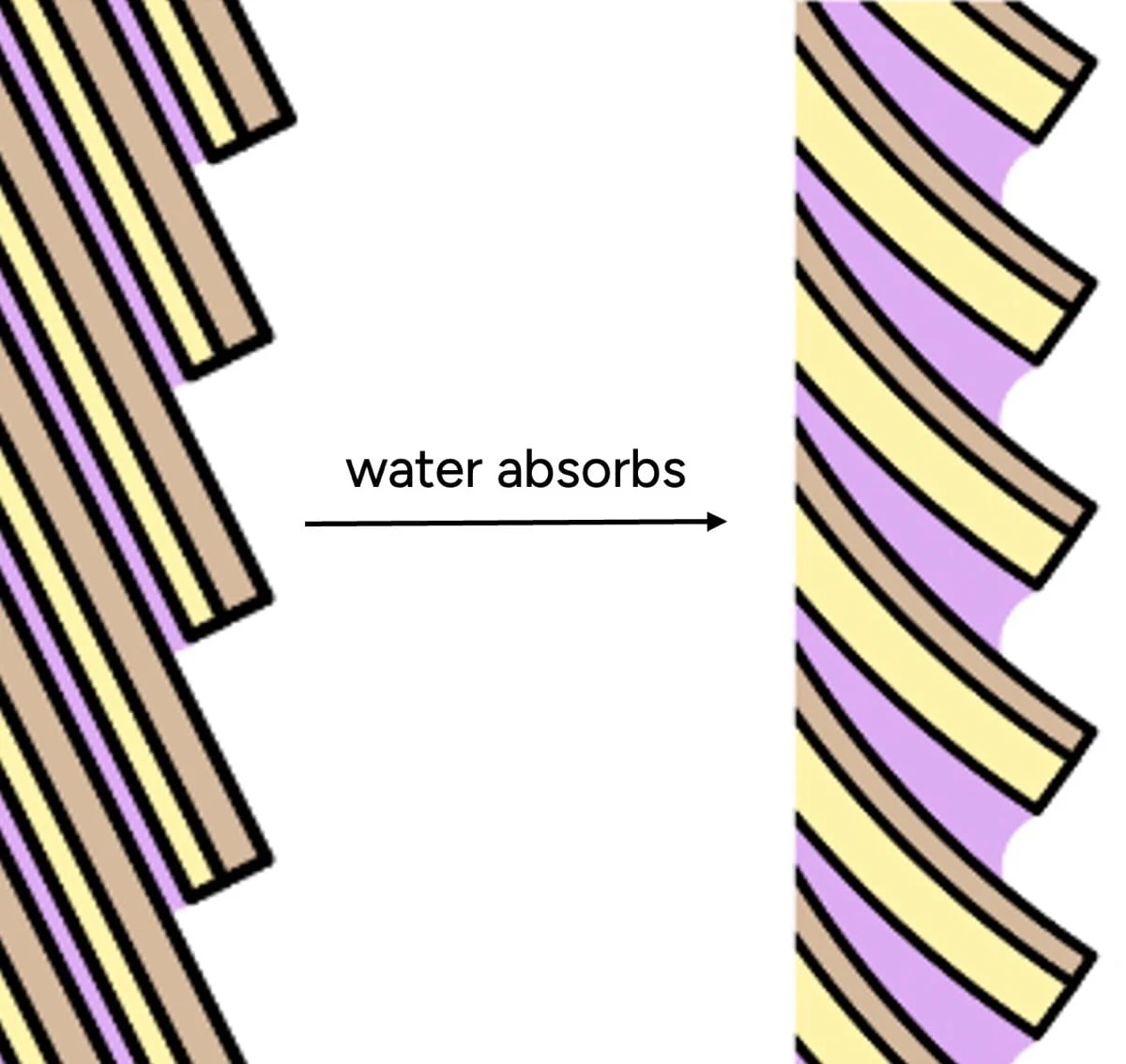
Imagine the protein chains as individuals, and their interactions as holding hands. In a dry state, these individuals are tightly linked, forming a robust network. Introduce water, and it’s akin to introducing many friendly new participants. The original individuals start holding hands with the newcomers (water molecules) instead of exclusively with each other. The result is a loosened, less cohesive network – wet hair with fewer internal bonds, making it weaker and more pliable. Hair can absorb up to 30% of its own weight in water, and this absorption causes the hair shaft to swell, leading to a measurable increase in diameter and a significant decrease in tensile strength, sometimes by as much as 30% to 50% compared to its dry state.
The Detangling Dilemma: Wet vs. Dry Combing
The impact of water on hair’s internal bonds has direct implications for everyday haircare, particularly detangling. The reduced internal bonding in wet hair makes it more elastic and stretchier. This increased elasticity might seem beneficial, but it also means wet hair is more susceptible to stretching beyond its elastic limit, leading to irreversible damage and breakage if excessive force is applied.
For straight hair, detangling when wet can be more damaging. The hair, though weaker, maintains its relatively straight form. Water also increases surface friction between strands, making them stick together more tightly. This combination of increased stickiness, reduced internal strength, and unchanged shape means more force is required to separate tangles, often resulting in breakage. Consequently, for straight hair types, detangling in a dry or slightly damp state with minimal tension is generally recommended.
Conversely, for curly and coily hair, wet detangling is often preferred and can be less damaging. While the hair is still weaker when wet, the intrinsic springiness and looser coil structure benefit from the lubricating and separating action of water, especially when combined with a conditioner. The water helps to soften the hair and allows strands to glide past each other more easily, reducing the overall resistance and force needed to remove tangles. The natural shape of curly hair means that trying to detangle it dry, where individual curls are often intertwined and brittle, can lead to much greater mechanical damage and frizz.
This "conundrum" extends even to humid environments, where hair partially absorbs moisture, making it weaker and more prone to tangling, especially for straight hair types. The study by Epps and Wolfram (1983) illustrated how the combing force required for hair can fluctuate significantly with changes in humidity, directly correlating with the hair’s water content. Effective detangling, regardless of hair type, hinges on minimizing force and friction, often best achieved with the assistance of emollients and lubricants found in conditioners.
The Tactile Illusion: Why More Water Feels "Worse"
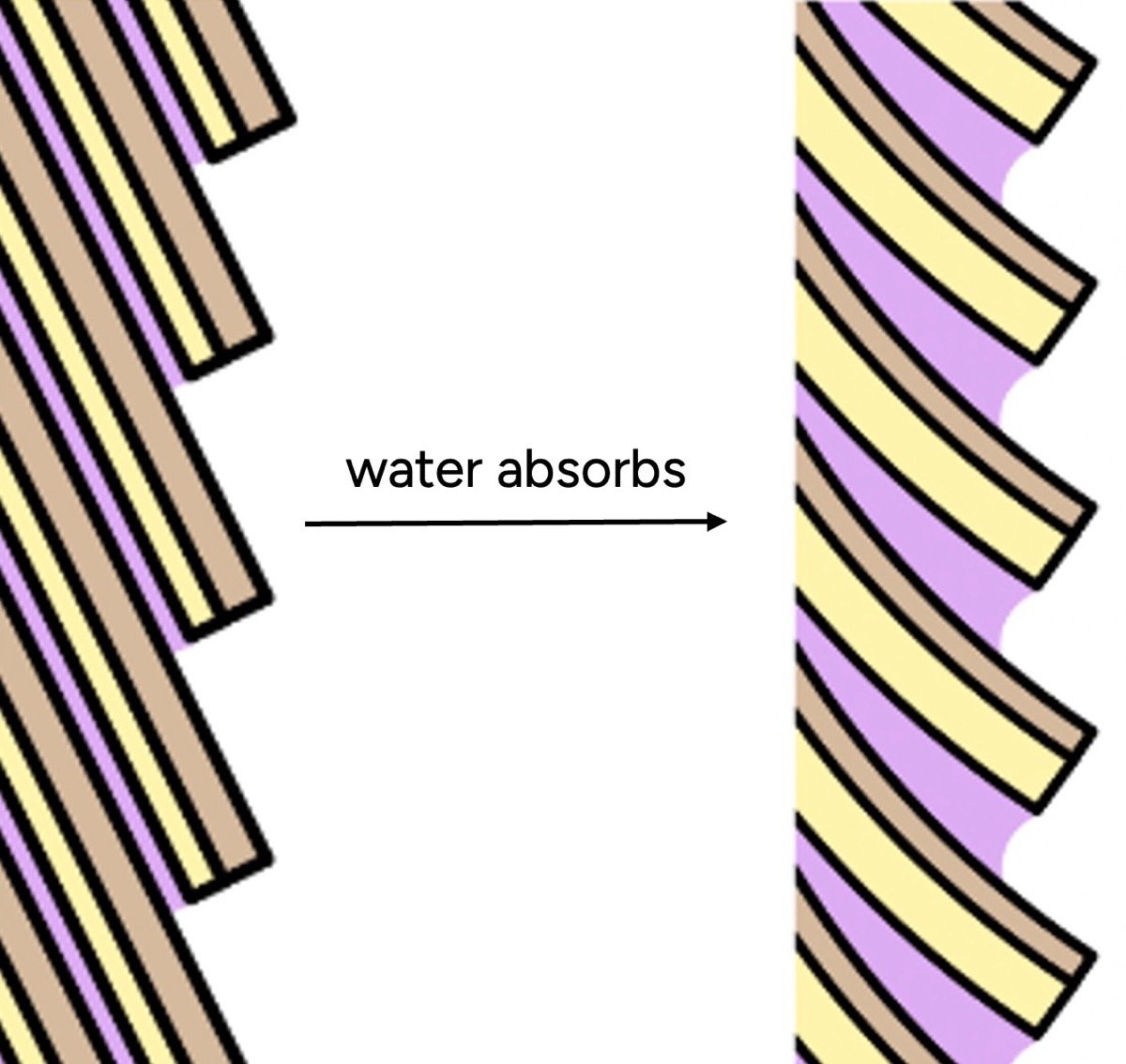
The perception that hair with more water feels "rougher" or "drier" stems from water’s interaction with the hair’s outermost layer: the cuticle. The cuticle consists of overlapping, scale-like cells that normally lie flat, creating a smooth, reflective surface. This smooth surface allows hair strands to glide past each other with minimal friction.
When hair absorbs water, the cuticle scales swell. However, this swelling is not uniform. The underside of each scale (the endocuticle) and the intercellular glue (the cell membrane complex) are more hydrophilic than the outer surface. This differential swelling causes the scales to lift and buckle, disrupting the smooth surface. The hair surface becomes microscopically uneven, creating a rougher texture.
Furthermore, water itself is inherently sticky due to its high surface tension and cohesive properties. This stickiness increases friction between hair strands. When we touch wet hair, our fingers encounter a bumpy, grippier surface, and the increased friction translates into a sensation we typically associate with "dryness" or "damage." This is a perceptual trick: our brains are accustomed to equating smoothness with health and moisture, and roughness with dryness. Hair, uniquely, often becomes rougher with increased water content at the surface, especially if that water is not managed or sealed in.
The True Mechanism of "Hydrating" Products
Given the scientific reality, how do products marketed as "hydrating" actually work? The answer lies not in adding more water to the hair’s internal structure, but primarily in conditioning its surface. Most "hydrating" shampoos, conditioners, and styling products contain ingredients like:
- Silicones: These polymers form a thin, smooth film over the cuticle, effectively "gluing" down lifted scales and reducing friction. They impart slip, shine, and a soft feel.
- Fatty Alcohols (e.g., Cetyl Alcohol, Stearyl Alcohol): These long-chain alcohols act as emollients, providing lubrication and a smooth feel to the hair surface.
- Emollients and Oils: Ingredients like argan oil, jojoba oil, or mineral oil can deposit on the hair surface, filling in microscopic gaps, smoothing the cuticle, and providing a protective barrier that reduces friction and helps manage water absorption/loss.
These ingredients work by mimicking the hair’s natural F-layer, an incredibly thin, hydrophobic lipid layer on the outermost cuticle that is gradually lost through daily wear and tear, washing, and chemical treatments. Conditioners effectively restore this protective, friction-reducing layer, making hair feel smoother, softer, and appear shinier – sensations that consumers interpret as "hydration." The focus is on surface modification and protection, not on significantly increasing the hair’s internal water content.
The Ritual of Washing: More Than Just Cleansing
The feeling of "reset" and improved manageability after a thorough wash and condition is a common experience, often attributed to "hydration." While water is central to this ritual, its benefits extend beyond simple moisture absorption:
- Optimized Ingredient Distribution: Water acts as an excellent solvent and spreading agent. During rinsing, it helps to evenly distribute conditioning ingredients across the hair shaft, ensuring comprehensive coverage and uniform surface modification. This is why rinse-off conditioners often feel more effective than leave-ins applied to dry hair.
- Temporary Bond Reset and Reshaping: Washing saturates the hair with water and often involves heat, which are powerful disruptors of temporary bonds. This "resets" the hair, releasing any lingering shapes from previous styling (e.g., heat-induced curls or straightening) and allowing the hair to return to its natural configuration as it dries. This highly pliable, "floppy" state makes hair more amenable to new styling and shaping.
- Enhanced Curl Clumping (for textured hair): For curly and coily hair, water’s surface tension plays a unique role. It encourages wet strands to clump together in parallel formations, a process known as "self-assembly." This leads to more defined, organized curls. When these larger, aligned clumps dry, they present a smoother, more uniform surface to the touch, and can even feel cooler due to better thermal conductivity, reinforcing the perception of "hydration."
- Microscopic Cuticle Realignment: Rapid drying, especially with high heat, or mechanical stretching can cause individual cuticle scales to buckle or distort. Soaking hair in water allows these scales to swell uniformly and then settle back into a flatter, more aligned configuration as the hair air dries or is dried gently. This microscopic realignment contributes to a smoother overall hair surface.
Crucially, these beneficial effects are primarily about water acting as a transient agent to facilitate shaping and product application, rather than its long-term retention within the hair fiber.

The Ideal Balance: Finding Hair’s "Sweet Spot"
Just like skin, hair thrives not at extremes of dryness or saturation, but within a balanced range of moisture content. There’s an "equilibrium moisture content" where hair exhibits optimal mechanical properties – neither too brittle nor too weak. This ideal range typically corresponds to the moisture levels found in ambient humidity.
Too Little Water (Very Dry Hair):
- Increased Static Electricity: In very low humidity, hair readily builds up static charge as it loses electrons through friction, leading to flyaways. Water, being conductive, helps to dissipate these charges.
- Brittleness and Fragility: When hair is excessively dry, its internal protein network is too rigid due to an abundance of temporary bonds. This lack of flexibility makes it unable to absorb mechanical stress (e.g., from brushing, combing, or styling) without forming microscopic cracks, which eventually lead to macroscopic breakage. The analogy of clipping dry vs. wet fingernails illustrates this perfectly: dry nails crack and split extensively, while wet nails, being more pliable, resist crack propagation.
Too Much Water (Excessively Wet Hair):
- Reduced Strength: As discussed, wet hair is significantly weaker and more prone to stretching and damage.
- Increased Swelling: While necessary for some processes, excessive and prolonged swelling can strain the hair’s structure.
- "Hygral Fatigue" (Myth Re-evaluated): The notion of "hygral fatigue," suggesting water itself damages hair through repeated wetting and drying, is largely considered a myth in its direct interpretation. Water itself is not inherently damaging. However, the processes associated with repeated wetting (e.g., increased friction during washing, vigorous towel drying, aggressive detangling of weakened wet hair) and the extreme fluctuations between swelling and shrinking can contribute to cumulative mechanical stress and damage over time. It’s not the water, but how hair is handled when wet, and the repeated physical changes, that can cause issues.
Strategies for Optimizing Hair’s Water Content and Feel
Achieving the "happy medium" for hair’s water content and its optimal feel involves a combination of product use and careful practices:
- Effective Conditioning: Regular use of conditioners is paramount. These products deposit emollients, silicones, and other film-forming agents that smooth the cuticle, reduce friction, and provide a protective barrier. They don’t necessarily "hydrate" in the sense of adding water internally, but they create the smooth, soft, and shiny appearance associated with healthy hair.
- Strategic Use of Oils: Certain oils can significantly impact hair’s interaction with water.
- Penetrating Oils (e.g., Coconut Oil): These oils can penetrate the hair shaft, reducing the amount of water absorbed and minimizing swelling. This can be beneficial for reducing hygral swelling and maintaining strength.
- Non-penetrating Oils (e.g., Jojoba, Argan, Mineral Oil): These oils primarily form a film on the hair surface, acting as occlusives. They reduce both water uptake and evaporation, helping to stabilize the hair’s moisture content and provide lubrication.
- Humectants (e.g., Glycerin, Hyaluronic Acid): These ingredients attract and hold water from the atmosphere. In moderately humid environments, they can draw moisture to the hair surface, keeping it supple. However, in very dry climates, humectants can paradoxically draw moisture out of the hair and into the drier air, potentially exacerbating dryness. Their effectiveness is highly dependent on environmental humidity.
- Heat Protectants: These products form a protective barrier on the hair surface, minimizing moisture loss and thermal damage during heat styling. They help ensure that any water present in the hair evaporates gently, preventing rapid and uneven drying that can lead to cuticle buckling.
- Gentle Handling and Drying Techniques: Minimizing mechanical stress, especially when hair is wet and vulnerable, is crucial. Using a wide-tooth comb with conditioner for detangling, blotting hair gently with a microfibre towel instead of vigorous rubbing, and allowing hair to air dry or using a diffuser on low heat can significantly reduce damage.
Conclusion: Embracing Scientific Nuance in Haircare
The relationship between water and hair is far more intricate than often portrayed. Water is neither a universal elixir nor a destructive force; it is a fundamental component and a powerful tool in haircare. Its ability to soften hair’s structure and reset its shape can be advantageous, particularly for distributing conditioning agents and defining curly patterns. However, its capacity to weaken hair’s internal bonds and roughen its surface demands careful management.
For consumers seeking hair that feels smooth, soft, and appears healthy, the focus should shift from the often-misleading concept of "hydration" to effective conditioning and gentle handling. Products that claim to "hydrate" primarily work by improving the hair’s surface smoothness and manageability through emollients and film-formers, not by dramatically increasing internal water levels. The notion of "hygral fatigue" as direct water damage is a misnomer; rather, it is the cumulative mechanical stress from handling hair in its weakened, swollen state, combined with extreme drying cycles, that contributes to long-term damage. By understanding these scientific principles, consumers can make more informed choices, cultivate healthier hair habits, and navigate the beauty landscape with greater clarity, ultimately achieving the desired aesthetic and tactile results without falling prey to misleading marketing.