Major depressive disorder (MDD) stands as a formidable global health crisis, ranking among the principal contributors to worldwide disability. Despite the availability of various therapeutic interventions, a significant subset of individuals, approximately 30%, develop treatment-resistant depression (TRD). This challenging condition is characterized by a persistent lack of sufficient symptom improvement with conventional antidepressant medications, leaving millions grappling with debilitating and often life-altering symptoms. In recent years, ketamine has emerged as a beacon of hope, demonstrating remarkable rapid antidepressant effects for those battling TRD. However, a critical barrier to its broader and more refined application has been the incomplete understanding of its precise mechanisms of action within the human brain. This knowledge gap has hindered the development of personalized treatment strategies, essential for optimizing patient outcomes.
A groundbreaking study, published on March 5, 2026, in the esteemed journal Molecular Psychiatry, has taken a significant stride toward demystifying ketamine’s therapeutic action. Spearheaded by Professor Takuya Takahashi of the Department of Physiology at Yokohama City University Graduate School of Medicine in Japan, the research team employed an innovative positron emission tomography (PET) imaging technique. This advanced methodology allowed for the direct observation of changes in the glutamate $alpha$-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptor (AMPAR). AMPARs are crucial protein complexes that orchestrate synaptic plasticity and glutamatergic signaling, fundamental processes for communication between neurons and vital for mood regulation.
"Although ketamine has shown rapid antidepressant effects in patients with treatment-resistant depression, its molecular mechanism in the human brain has remained unclear," stated Professor Takahashi, underscoring the long-standing challenge his team sought to address. "Our research aimed to provide direct, in-vivo evidence of how ketamine influences these critical brain receptors and how these changes translate into clinical improvement."
Visualizing Brain Receptors with a Novel PET Tracer
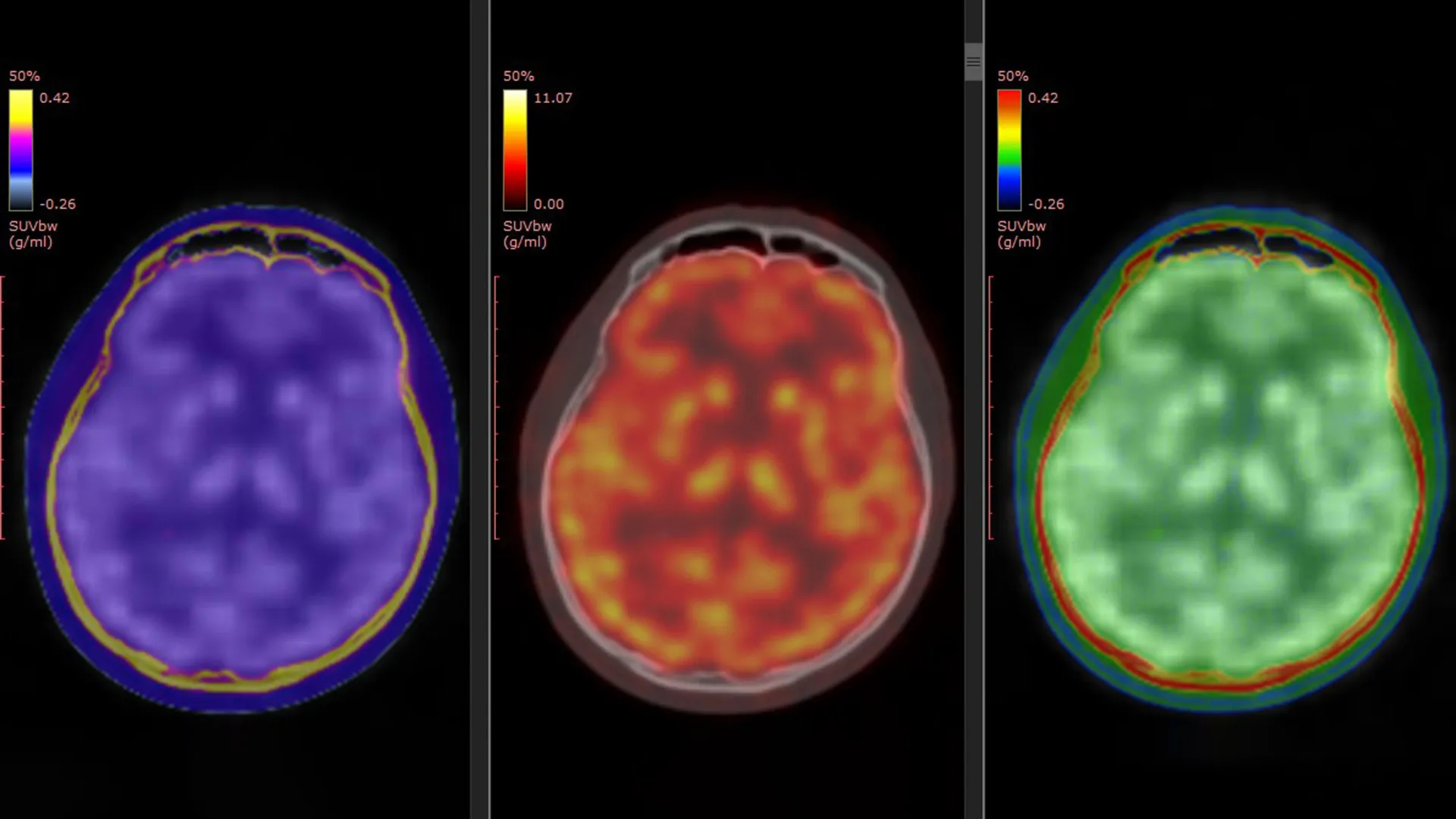
The linchpin of this investigation was a novel PET tracer, designated [¹¹C]K-2, developed by Professor Takahashi’s group. This sophisticated tracer possesses the unique capability to visualize cell-surface AMPARs directly within the living human brain. Previous preclinical studies, conducted in laboratory settings and animal models, had strongly suggested a role for AMPAR activity in mediating ketamine’s antidepressant effects. However, the current study marks the first instance of providing direct, empirical evidence of this process occurring in human subjects, thereby bridging a critical gap between animal research and human clinical application.
To achieve this crucial insight, the researchers meticulously compiled and analyzed data from three registered clinical trials that had been previously conducted in Japan. The study cohort comprised 34 patients formally diagnosed with treatment-resistant depression and 49 healthy individuals who served as a control group, providing a vital baseline for comparison.
Participants diagnosed with TRD were administered either intravenous ketamine or a placebo over a two-week treatment period. A critical component of the study design involved PET brain imaging conducted at two key junctures: prior to the commencement of treatment and again following the final ketamine or placebo infusion. This temporal imaging approach enabled the researchers to precisely quantify and compare any alterations in AMPAR levels and their spatial distribution within the brain throughout the treatment course. This meticulous methodology was designed to capture the dynamic interplay between ketamine administration and its impact on neurobiological markers associated with depression.
Region-Specific Brain Changes Linked to Symptom Relief
The findings revealed a compelling pattern of AMPAR abnormalities in individuals with TRD when contrasted with their healthy counterparts. These differences were not uniformly distributed across the entire brain but were instead localized to specific, functionally distinct brain regions. This observation suggested that TRD might be associated with localized disruptions in glutamatergic neurotransmission rather than a global deficit.
Crucially, the study demonstrated that ketamine did not induce uniform changes in AMPAR density throughout the brain. Instead, the observed improvements in depressive symptoms were intimately linked to dynamic, region-specific adjustments in AMPAR levels. In certain cortical areas, there was an observable increase in AMPAR density, potentially reflecting enhanced neuronal communication and plasticity. Conversely, regions implicated in reward processing and motivation, notably the habenula, exhibited a reduction in AMPAR density. The researchers found a strong correlation between these region-specific shifts in AMPAR distribution and the degree of symptom relief experienced by the patients.
"Ketamine’s antidepressant effect in patients with TRD is mediated by dynamic changes in AMPAR in the living human brain," Professor Takahashi elaborated. "Using a novel PET tracer, [¹¹C]K-2, we were able to visualize how ketamine alters AMPAR distribution across specific brain regions and how these changes correlate with improvements in depressive symptoms."
These findings provide the first direct human evidence that substantiates mechanisms previously inferred from animal studies and establishes a concrete link between these molecular alterations and tangible clinical antidepressant effects. This validation is paramount for building confidence in ketamine as a therapeutic agent and for refining its application.
Potential Biomarker for Predicting Treatment Response
Beyond elucidating the intricate mechanisms of ketamine’s action, the study’s implications extend to practical clinical applications. The capacity to visualize AMPAR density via PET imaging holds significant promise as a potential biomarker. Such a biomarker could empower clinicians to more accurately assess and predict an individual patient’s likely response to ketamine treatment for TRD.
The challenge of identifying reliable biological markers for treatment response is a persistent and critical goal in mental health care, particularly given the substantial proportion of patients who do not achieve adequate relief from standard antidepressant therapies. The ability to predict treatment efficacy before initiating a potentially costly and time-consuming course of therapy could revolutionize the management of TRD, saving valuable time and resources for patients and healthcare systems alike.
Toward More Personalized Depression Treatments
By enabling scientists to directly observe AMPAR activity in the living human brain, this pioneering research effectively bridges a long-standing chasm between fundamental laboratory investigations and the realities of clinical psychiatry. The study unequivocally identifies AMPAR modulation as a central mechanistic pathway underlying ketamine’s rapid antidepressant effects. Furthermore, it strongly suggests that AMPAR PET imaging could serve as a powerful tool to guide the development of more personalized and tailored treatment strategies for individuals suffering from treatment-resistant depression.
The implications of this research are profound. It paves the way for a future where treatment decisions for TRD are informed by objective biological data, moving beyond a trial-and-error approach. This could lead to earlier identification of effective treatments, reduced patient suffering, and improved overall quality of life for those affected by this debilitating condition. Ultimately, this work represents a significant leap forward in the quest for more precise and effective therapies for people living with the profound challenges of treatment-resistant depression.
The research was made possible through substantial funding from various governmental and private institutions. Key support was provided by the Ministry of Education, Culture, Sports, Science and Technology (through Special Coordination Funds for Promoting Science and Technology); the Japan Agency for Medical Research and Development (AMED) under grant numbers JP18dm0207023, JP19dm0207072, JP24wm0625304, JP25gm7010019, and JP20dm0107124; the Japan Society for the Promotion of Science KAKENHI, with grants including 22H03001, 20H00549, 20H05922, 23K10432, 19H03587, 20K20603, 22K15793, and 21K07508; the Takeda Science Foundation; the Keio Next-Generation Research Project Program; the SENSHIN Medical Research Foundation; and the Japan Research Foundation for Clinical Pharmacology. This broad spectrum of support highlights the collaborative and multi-faceted nature of advancing complex neuroscientific research.
The prevalence of major depressive disorder is estimated to affect over 280 million people globally, underscoring the immense public health burden it represents. While the exact pathophysiology of TRD remains elusive, it is thought to involve a complex interplay of genetic, environmental, and neurobiological factors, including dysregulation in neurotransmitter systems like the glutamatergic system. The development of ketamine, initially used as an anesthetic, into a rapid-acting antidepressant agent was itself a significant paradigm shift in psychiatric treatment. However, its rapid onset of action, often within hours, contrasted sharply with the weeks or months typically required for conventional antidepressants to take effect. This rapid efficacy fueled intense scientific inquiry into its underlying biological mechanisms.
The timeline leading to this publication reflects a progression of scientific understanding. Initial observations of ketamine’s antidepressant potential in the late 1990s and early 2000s, primarily in anecdotal reports and small clinical trials, spurred further investigation. The subsequent identification of the NMDA receptor as a primary target of ketamine in the brain laid the groundwork for understanding its acute effects. However, it became increasingly clear that ketamine’s influence extended beyond NMDA receptors, with evidence pointing towards AMPA receptor potentiation and downstream effects on synaptic plasticity as critical components of its sustained antidepressant action. The development of specific PET tracers like [¹¹C]K-2 represents the culmination of years of radiochemistry and molecular imaging research, enabling the direct visualization and quantification of these receptor dynamics in living humans. This study, published in 2026, represents a significant milestone, providing the empirical human data that validates these hypothesized mechanisms.
The implications for pharmaceutical development are also substantial. A deeper understanding of AMPAR modulation could lead to the development of novel compounds that target this pathway more specifically, potentially offering improved efficacy and fewer side effects compared to ketamine. Furthermore, the identification of AMPAR density as a potential predictive biomarker could inform the design of future clinical trials, allowing researchers to stratify patient populations based on their likely response, thereby increasing the efficiency and success rate of drug development for TRD.
The broader impact of this research extends to patient advocacy and mental health policy. By demystifying the biological underpinnings of effective treatments like ketamine, it can help reduce stigma associated with mental illness and foster greater public trust in scientific advancements in psychiatry. It also strengthens the case for increased investment in research aimed at understanding and treating complex mental health conditions. The ability to offer more precise and personalized treatments for TRD could significantly alleviate the immense personal suffering and societal cost associated with this condition, offering renewed hope for millions worldwide.