The widespread concept of "hair porosity" has become a cornerstone of modern haircare advice, influencing countless product choices and routines. However, a closer examination of the science reveals that many popular DIY tests designed to determine hair porosity are fundamentally flawed, leading consumers and even some professionals down a path of misinterpretation. These tests, while often correlating with positive haircare outcomes, do not actually measure the internal porosity of hair but rather its surface interaction with water, primarily governed by the principles of surface tension and the integrity of the hair’s outermost layer.
The Rise of Porosity in Haircare and its Misconceptions
In recent years, the term "hair porosity" has surged in popularity, particularly within online beauty communities and specialized haircare niches, such as the natural and curly hair movements. The premise is that understanding whether hair has "low," "medium," or "high" porosity allows individuals to select products and techniques best suited for their hair type. Low porosity hair is often described as having tightly bound cuticles that resist moisture absorption but retain it well, while high porosity hair is characterized by open or damaged cuticles that readily absorb moisture but struggle to retain it. This framework promised a personalized approach to haircare, moving beyond generic recommendations.
The allure of this concept led to the proliferation of simple, at-home tests like the "Float Test" and the "Drop Test." The Float Test involves placing a single strand of hair into a glass of water, with the theory that sinking hair indicates high porosity (absorbing water and becoming denser) and floating hair indicates low porosity (repelling water). The Drop Test suggests that water drops bead up on low porosity hair but flatten and absorb quickly into high porosity hair. While these tests appear intuitive, their interpretations are rooted in a scientific misunderstanding of how hair interacts with water.
Deconstructing the "Waterproof Hair" Myth

A central misconception underlying the popular porosity narrative is the idea that healthy, undamaged hair is essentially "waterproof." Proponents of this myth suggest that a pristine cuticle layer acts as an impermeable barrier, sealing out water. Conditioners, in this view, are thought to mimic this sealing function for damaged hair. However, scientific research consistently debunks this notion.
Undamaged, conditioned hair is far from waterproof; it possesses a significant capacity for water absorption. Studies, such as those detailed in C.R. Robbins’ seminal work, Chemical and Physical Behavior of Human Hair, demonstrate that human hair can absorb a substantial amount of water, often up to 30% of its own weight, within minutes when exposed to high humidity. The water content of hair is dynamic, rapidly adjusting to changes in ambient humidity.
For instance, the following data illustrates the direct relationship between relative humidity and the weight of water absorbed by hair:
| Relative humidity (%) | Weight of water absorbed (%) |
|---|---|
| 0 | 0 |
| 8 | 3.9 |
| 40 | 10.2 |
| 63 | 14.8 |
| 86 | 22.6 |
| 100 | 31.2 |
Source: Robbins CR. Chemical and Physical Behavior of Human Hair. Springer Berlin Heidelberg 2012.
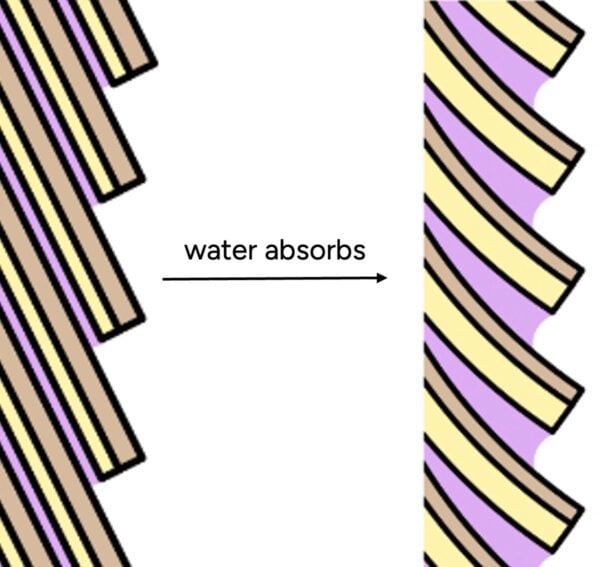
This data clearly shows that hair readily takes on water, even at moderate humidity levels. The mechanism behind this absorption lies in the hair’s microscopic structure. The cuticle, often visualized as overlapping scales, is not a perfectly sealed unit. Instead, it resembles a pinecone, with inherent gaps and overlaps. Furthermore, the hair’s natural conditioning F-layer, a thin lipid layer rich in 18-methyleicosanoic acid (18-MEA), primarily covers the top of each cuticle scale, leaving ample spaces for water molecules to penetrate.
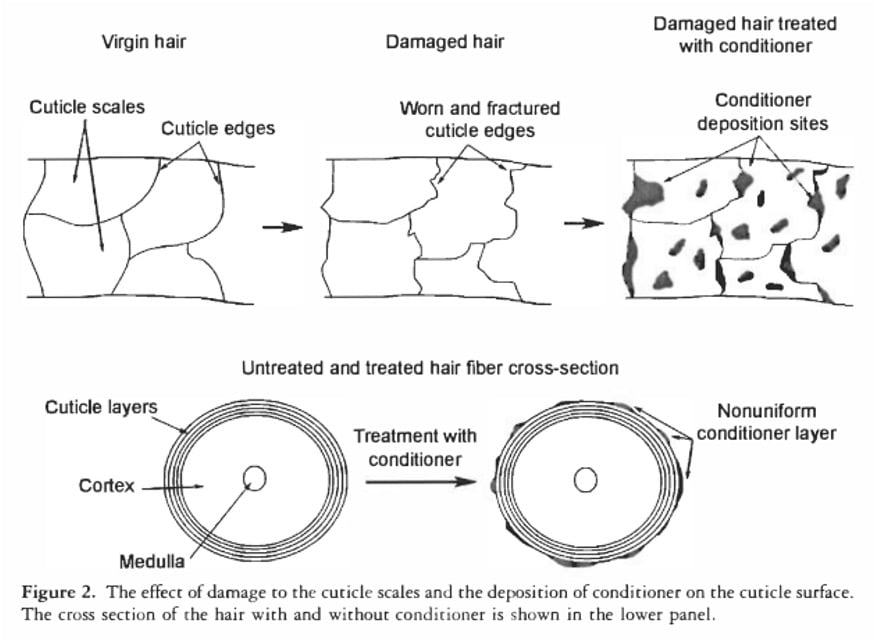
Even synthetic conditioners, which are designed to smooth the hair’s surface and improve manageability, do not create an impermeable barrier. Research by La Torre and Bhushan (2006) using atomic force microscopy shows that conditioners deposit on hair in "blobs" rather than forming a continuous, uniform film. While these microscopic blobs are sufficient to make hair feel smoother and softer to the touch, individual water molecules are infinitesimally small and can easily navigate around these deposits. Therefore, conditioners enhance the surface properties of hair, reducing friction and improving aesthetics, but they do not render hair waterproof or significantly impede water absorption at the molecular level.
The Flawed Foundations: Analyzing Popular Porosity Tests
Given that undamaged hair is not waterproof and readily absorbs water, the interpretations offered by the Float Test and Drop Test become highly questionable. These tests, instead of revealing internal porosity, are primarily demonstrating how water interacts with the surface of the hair.
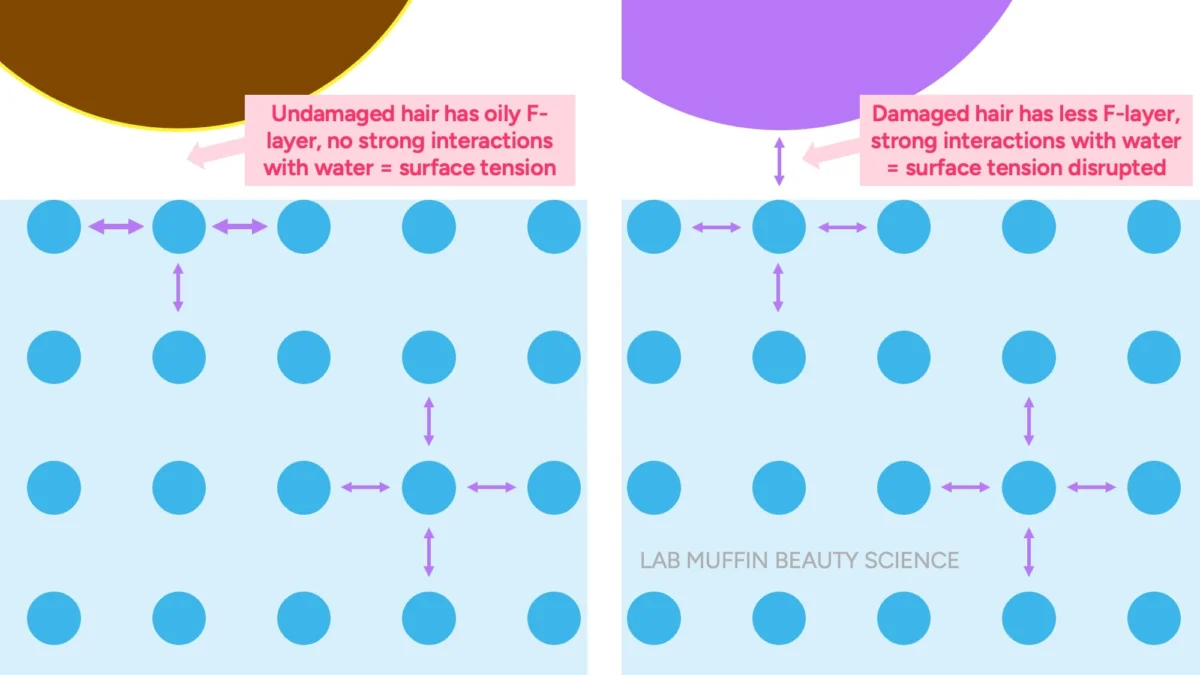
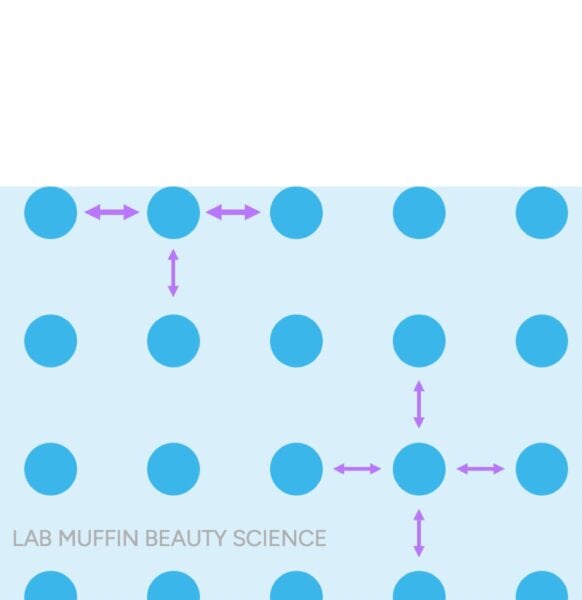
The Float Test Debunked: The Float Test posits that hair that sinks quickly is "high porosity" because it has absorbed water, while hair that floats is "low porosity" due to its resistance to water. The reality is more nuanced. Hair, regardless of its "porosity," is denser than water and should eventually sink. The phenomenon of hair floating is not due to its inherent resistance to internal water absorption, but rather to the principle of surface tension.
When undamaged hair, coated with its natural F-layer, is placed on water, the hydrophobic (water-repelling) surface of the hair minimizes interaction with the water molecules. This allows the strong cohesive forces of water molecules at the surface – the very definition of surface tension – to form a "skin" that can support the hair strand. Much like a tiny insect or a carefully placed paperclip can float on water despite being denser, the hair is supported by this invisible barrier. Damaged hair, however, has often lost significant portions of its F-layer, exposing a more hydrophilic (water-attracting) surface. When this damaged, hydrophilic surface meets water, it forms hydrogen bonds with the water molecules, effectively disrupting the surface tension. With the surface tension compromised, the hair’s natural density causes it to sink. Thus, the Float Test is not measuring how much water the hair absorbs internally, but rather the integrity of its hydrophobic surface layer.
The Drop Test Debunked: Similarly, the Drop Test, which observes how a drop of water behaves on a strand or lock of hair, is also explained by surface interactions rather than internal absorption. The test claims that water beading on hair indicates low porosity, while water flattening and spreading indicates high porosity. The explanation given is that "high porosity" hair, being "full of holes," absorbs the water quickly.
However, as established, even undamaged hair can absorb around 30% of its weight in water, and damaged hair might absorb slightly more, perhaps up to 45%. This difference is not significant enough to explain the immediate flattening and spreading observed in the Drop Test. The key factor here is also surface tension and wettability. On undamaged hair with an intact hydrophobic F-layer, the water drop minimizes contact with the hair surface due to poor adhesion, maintaining its spherical shape (beading) because its internal cohesive forces (surface tension) are stronger than its adhesive forces to the hair. On damaged hair, where the F-layer is compromised and the surface is more hydrophilic, water molecules adhere more strongly to the hair. This increased adhesion overcomes the water’s surface tension, causing the drop to flatten out and spread along the hair surface and wick between individual strands, creating the illusion of rapid absorption. The water is spreading on and between the hair fibers, not necessarily soaking into them more quickly in its liquid state.
Understanding Surface Tension: The Real Science
To truly grasp why these tests are misleading, a deeper dive into surface tension is crucial. Surface tension is a property of the surface of a liquid that allows it to resist an external force. It is caused by the cohesive forces between liquid molecules, particularly hydrogen bonds in water. Inside a body of water, each water molecule is surrounded and pulled equally in all directions by its neighbors through hydrogen bonds. However, at the surface, water molecules have fewer neighbors above them. This imbalance causes the surface molecules to be pulled inward, towards the bulk of the liquid, and also sideways, along the surface. This inward and sideways pull creates a net inward force, resulting in the liquid’s surface behaving like an elastic "skin" or membrane.
This "skin" is strong enough to support objects that are denser than water, as long as they do not disrupt the surface’s integrity. Classic examples include insects walking on water, or a carefully placed paperclip floating. A paperclip, made of steel, is approximately eight times denser than water. Yet, it can float if placed gently, demonstrating that its ability to stay afloat is due to surface tension, not its density or ability to absorb water (which it cannot). If one were to disrupt this surface tension – by poking the paperclip or adding a surfactant like detergent – the paperclip would immediately sink.
Applying this to hair, the F-layer on healthy hair provides a low-energy, hydrophobic surface that allows water’s surface tension to remain largely intact, enabling the hair to "float" on this invisible skin. When the F-layer is damaged or removed, the hair surface becomes higher energy and more hydrophilic, allowing water molecules to adhere more readily, thus breaking the surface tension and causing the hair to sink. This is the direct analogy to the paperclip and detergent.
Hair Hydration: Gas vs. Liquid Water Absorption
It’s important to distinguish between how hair absorbs water from humidity (gaseous water) versus how it interacts with liquid water. In humid air, individual water molecules exist as a gas, meaning they are not bound together by the strong hydrogen bonds found in liquid water. These tiny, unassociated gas molecules are small enough to easily "wiggle" their way between the cuticle scales and into the hair’s cortex without encountering the resistance of surface tension. This explains why hair can rapidly absorb a significant percentage of its weight in water from the air, irrespective of its perceived "porosity" from a float test.

Conversely, liquid water’s interaction with the hair surface is dominated by surface tension. While hair can and does absorb liquid water internally, the initial observed behavior in tests like the Float and Drop Tests is primarily a surface phenomenon. The rate and extent of liquid water absorption into the hair’s cortex are influenced by the degree of cuticle damage, but the initial interaction—whether it floats, sinks, beads, or spreads—is a function of the hair’s surface chemistry and its effect on water’s surface tension.
Why the "Porosity" Advice Still Seems Effective
The paradox is that despite the scientific inaccuracies of these DIY tests, the haircare advice often derived from them frequently leads people to products that do seem to suit their hair better. This is largely a coincidence, stemming from the fact that the tests, while mislabeled, do indirectly assess a critical aspect of hair health: surface damage.
When a Float Test or Drop Test yields a "high porosity" result, it typically means the hair’s F-layer is compromised, indicating surface damage (e.g., from chemical treatments, heat styling, mechanical stress). Hair with significant surface damage often benefits from products recommended for "high porosity" hair, such as heavier conditioners, protein treatments, and occlusive agents, because these products help to smooth the cuticle, provide temporary repair, and reduce moisture loss from the damaged internal structure. These products address the symptoms of damage, which happen to correlate with the "high porosity" label.
Conversely, hair that performs as "low porosity" in these tests often has a relatively intact F-layer and less surface damage. Such hair might indeed be prone to product buildup and could benefit from lighter products and clarifying shampoos, which are often recommended for "low porosity" hair. This correlation further reinforces the perceived validity of the tests, even though the underlying scientific explanation for the test results is incorrect. The advice works because it aligns with the actual condition of the hair’s surface, not because it accurately diagnoses an internal porosity level in the way the term implies.
Implications for Consumers: Beyond the Lie

For consumers seeking to understand and care for their hair effectively, the debunking of popular porosity tests means shifting focus from a misleading label to a more holistic assessment of hair health. Instead of relying on a float test, individuals should observe their hair’s actual behavior:
- Visual Inspection: Does the hair look dull or shiny? Are there split ends or signs of breakage?
- Tactile Assessment: Does it feel rough or smooth? Is it easily tangled or slippery?
- Product Response: How does the hair react to different types of products? Does it feel dry after using lightweight conditioners, or weighed down by heavy ones? Does it absorb styling products quickly or do they sit on the surface?
- Elasticity: Does a wet strand stretch a little before breaking, or does it snap immediately?
- Professional Consultation: A qualified hairdresser or trichologist can provide a more accurate assessment of hair condition and needs.
Focusing on the hair’s overall condition—whether it’s damaged, dry, oily, fine, coarse, straight, or curly—provides a more practical and scientifically sound basis for choosing haircare products and routines. Understanding that hair naturally absorbs moisture and that its surface integrity is key to its appearance and feel is far more useful than an inaccurate "porosity" label.
Professional Perspective: A Warning for Hairdressers
The implications of these findings are particularly critical for haircare professionals. While the "porosity" framework might be useful for general product recommendations, relying on these superficial tests to guide technical procedures like chemical treatments (coloring, perms, relaxers) can lead to serious errors and potential damage.
For example, if a hairdresser relies on a Float Test to determine that a client has "high porosity" hair, they might mistakenly assume that chemical treatments will absorb rapidly and reduce processing time. However, as established, the test primarily indicates surface damage. While damaged hair might sometimes absorb chemicals more quickly due to a compromised cuticle, this is not a universal rule and depends heavily on the specific type and extent of damage. Some forms of damage might actually impede chemical penetration, or the hair might be so compromised that a standard treatment would cause severe breakage.
Leading cosmetic chemists and hair scientists, including Dr. Michelle Wong of Lab Muffin Beauty Science, strongly advise against using these popular porosity tests for professional chemical treatment timing. The most reliable and responsible method for hairdressers remains direct strand testing. Applying the chemical product to a small, inconspicuous strand of the client’s hair and carefully monitoring its reaction provides real-time, accurate information on processing time, ensuring optimal results and minimizing damage.
The Broader Impact: Science Communication in Beauty
The persistence of the hair porosity myth highlights a broader challenge in the beauty industry: the gap between scientific understanding and popular consumer narratives. In an age of rapid information dissemination through social media, scientifically inaccurate concepts can quickly gain traction, often fueled by anecdotal evidence and simplified explanations.
The work of science communicators like Dr. Wong, who translate complex scientific principles into accessible language, is invaluable in dispelling these myths. By emphasizing evidence-based approaches and explaining the underlying chemistry and physics of hair, they empower consumers to make more informed decisions and encourage professionals to adopt best practices. This movement towards greater scientific literacy in beauty not only improves individual haircare outcomes but also elevates the industry as a whole, fostering a more transparent and credible environment.
In conclusion, while the intention behind hair porosity tests may be to personalize haircare, their scientific foundation is weak. They do not accurately measure internal porosity but rather the surface condition of the hair and its interaction with water’s surface tension. Recognizing this distinction allows for a more effective and scientifically sound approach to haircare, one that prioritizes understanding actual hair damage and needs over misleading labels.
References
Robbins CR. Chemical and Physical Behavior of Human Hair. 5th ed. Springer Berlin Heidelberg 2012.

La Torre C, Bhushan B. Nanotribological effects of silicone type, silicone deposition level, and surfactant type on human hair using atomic force microscopy. J Cosmet Sci. 2006;57(1):37-56.