The pervasive notion that hair requires "hydration" to be healthy, as suggested by a multitude of products, presents a complex and often misunderstood aspect of haircare science. While many consumers and even some industry professionals readily assent to the idea that water is unequivocally beneficial for hair, a deeper dive into the molecular interactions reveals a more nuanced reality. The scientific community, including leading cosmetic scientists, often grapples with the challenge of correcting ingrained public perceptions, sometimes even conceding to market demands over strict scientific accuracy to avoid alienating consumers. This article aims to disentangle the myths from the verifiable science, offering a comprehensive understanding of how water truly interacts with human hair and what this means for effective haircare.
Unpacking the "Hydrated Hair" Misconception
For decades, the beauty industry has championed "hydrating" products, leading consumers to associate increased water content with softness, smoothness, and overall hair health. This marketing narrative, while effective in sales, stands in stark contrast to the findings of rigorous scientific inquiry. In reality, hair that feels soft, smooth, and appears "healthy" often possesses a lower intrinsic water content compared to hair perceived as rough, "dry," or damaged. This counterintuitive finding was underscored by a significant study where participants consistently rated hair with less water as more moisturized and less damaged. The discrepancy arises from a fundamental misunderstanding of water’s physical effects on hair structure and surface texture.
The Microscopic World: Hair Structure and Water’s Influence
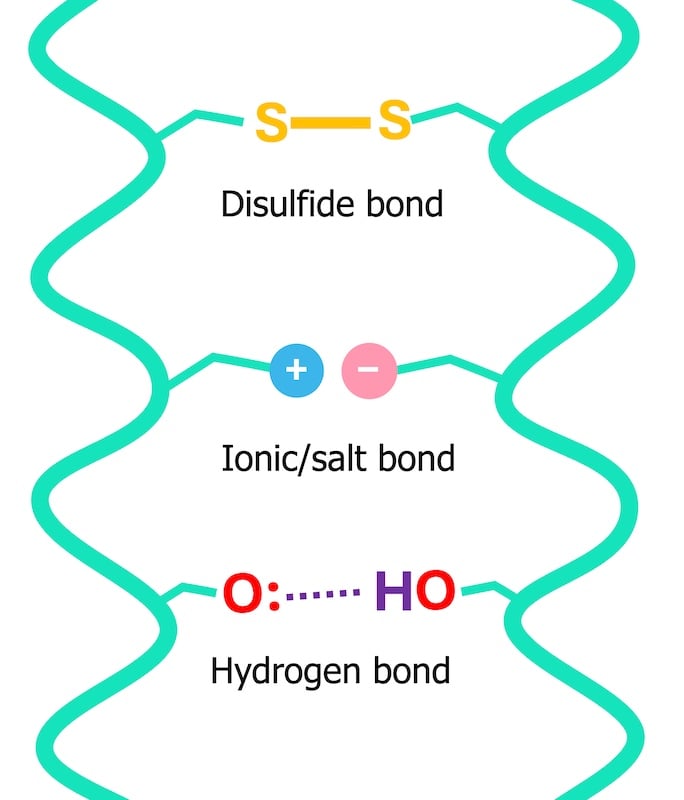
To comprehend water’s role, one must first appreciate the intricate architecture of a hair strand. Hair is primarily composed of keratin proteins, which are bound together by various types of chemical linkages, providing both strength and shape. These bonds fall into two main categories:
- Permanent bonds (disulfide bonds): These are strong covalent bonds that largely dictate the hair’s natural curl pattern and are broken only by chemical processes like perming or relaxing.
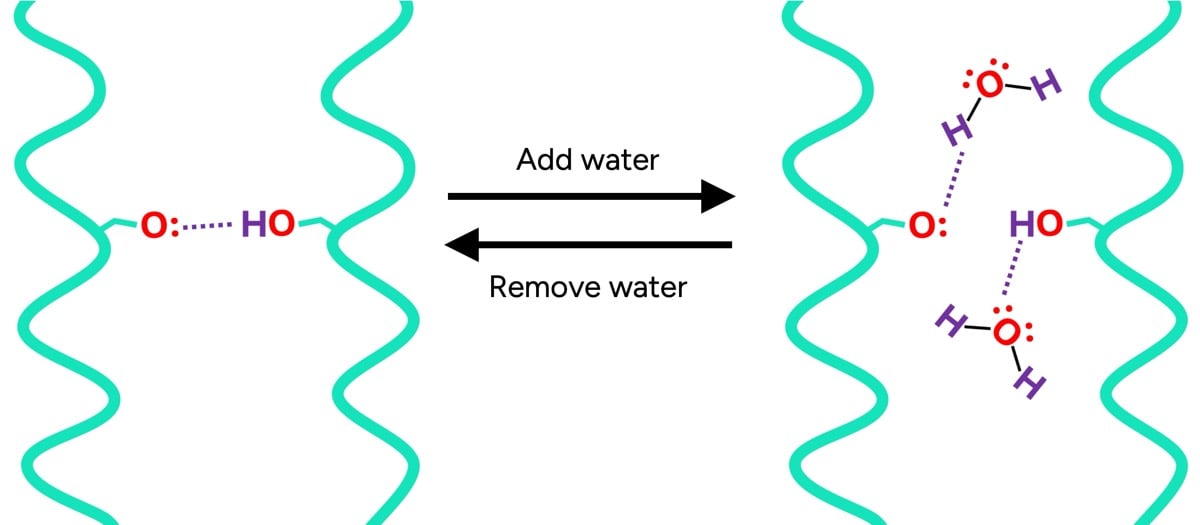
- Temporary bonds (hydrogen and ionic bonds): These are weaker, non-covalent interactions that are highly sensitive to environmental factors, particularly water and heat. They are responsible for maintaining the hair’s immediate shape and contribute significantly to its mechanical properties.
The inner core of the hair, known as the cortex, is particularly susceptible to water. In dry hair, temporary bonds form between adjacent protein chains, effectively "holding hands" at a molecular level, contributing to the hair’s rigidity. However, upon exposure to water, these protein-protein bonds are readily disrupted. Water molecules, being polar, interpose themselves between the protein chains, forming new hydrogen bonds with the proteins. This phenomenon, akin to a molecular "dance-off," results in fewer protein-protein bonds, rendering wet hair significantly weaker and more elastic.
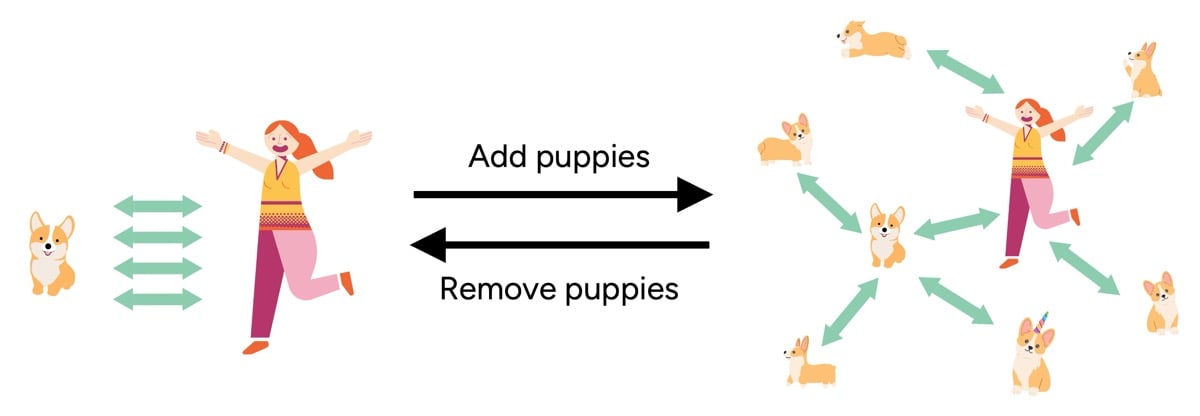
An apt analogy illustrates this molecular interaction: imagine two dogs, you and Felix, interacting closely in an empty park. Introduce a multitude of friendly puppies (water molecules), and both you and Felix will naturally interact more with the newcomers, reducing your direct interaction with each other. When the "puppies" (water) leave, the original bond strengthens again. Similarly, wet hair has fewer stabilizing internal bonds, making it more pliable but also more vulnerable to damage from mechanical stress.
Water’s Dual Nature: Detangling and Mechanical Integrity
The impact of water on hair’s temporary bonds directly influences its mechanical properties, affecting aspects like stiffness, elasticity, and susceptibility to damage during detangling. Dry hair, with its higher density of internal bonds, is stiffer and more brittle. Wet hair, conversely, is stretchier and more elastic due due to the disruption of these bonds.
This difference creates a fascinating conundrum for detangling:
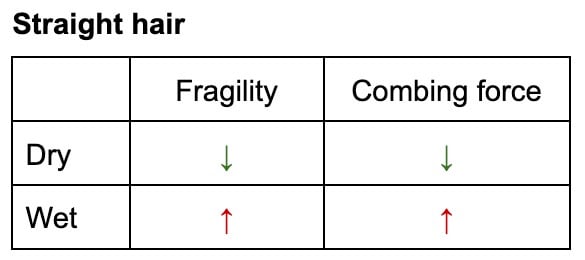
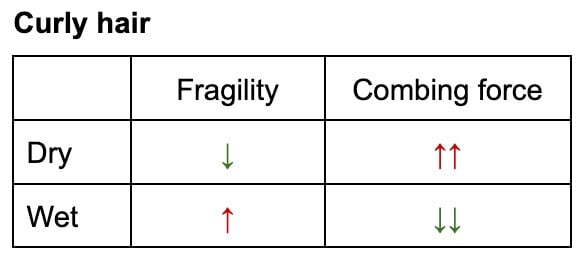
- Straight Hair: For straight hair, detangling when wet can be more damaging. Although more elastic, the weakened internal structure combined with increased friction between wet, clinging strands can lead to greater breakage. The hair retains its original shape but is fundamentally weaker, and the water acts as an adhesive, increasing resistance.
- Curly Hair: Paradoxically, for curly hair, wet detangling can be less damaging. Despite the temporary weakening, the relaxed, looser curl pattern in its wet state reduces resistance and tangling. The overall force required to detangle is lower, mitigating the risk of breakage. This is often why curly hair routines emphasize detangling in the shower with conditioner.
This nuanced effect extends beyond fully wet hair to humid environments. Studies, such as that by Epps and Wolfram, have demonstrated that even elevated humidity can alter hair’s mechanical response, influencing the force required for combing and the potential for damage. The implication is clear: the optimal detangling strategy is not universal but depends on hair type and condition, prioritizing methods that minimize mechanical force. Conditioner plays a crucial role in both wet and dry detangling, providing lubrication that reduces friction and minimizes damage.
The Sensory Deception: Why More Water Can Feel "Worse"
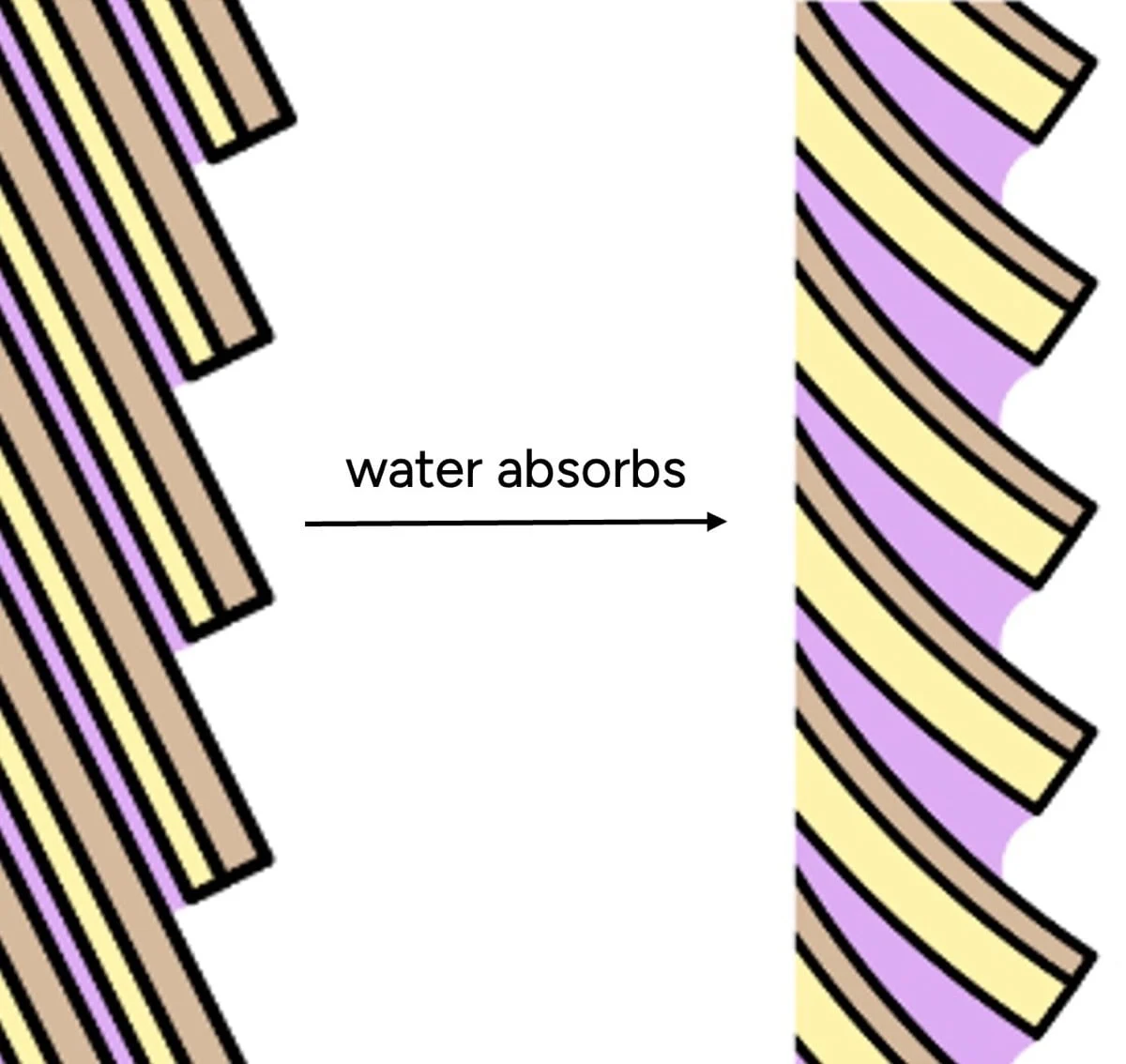
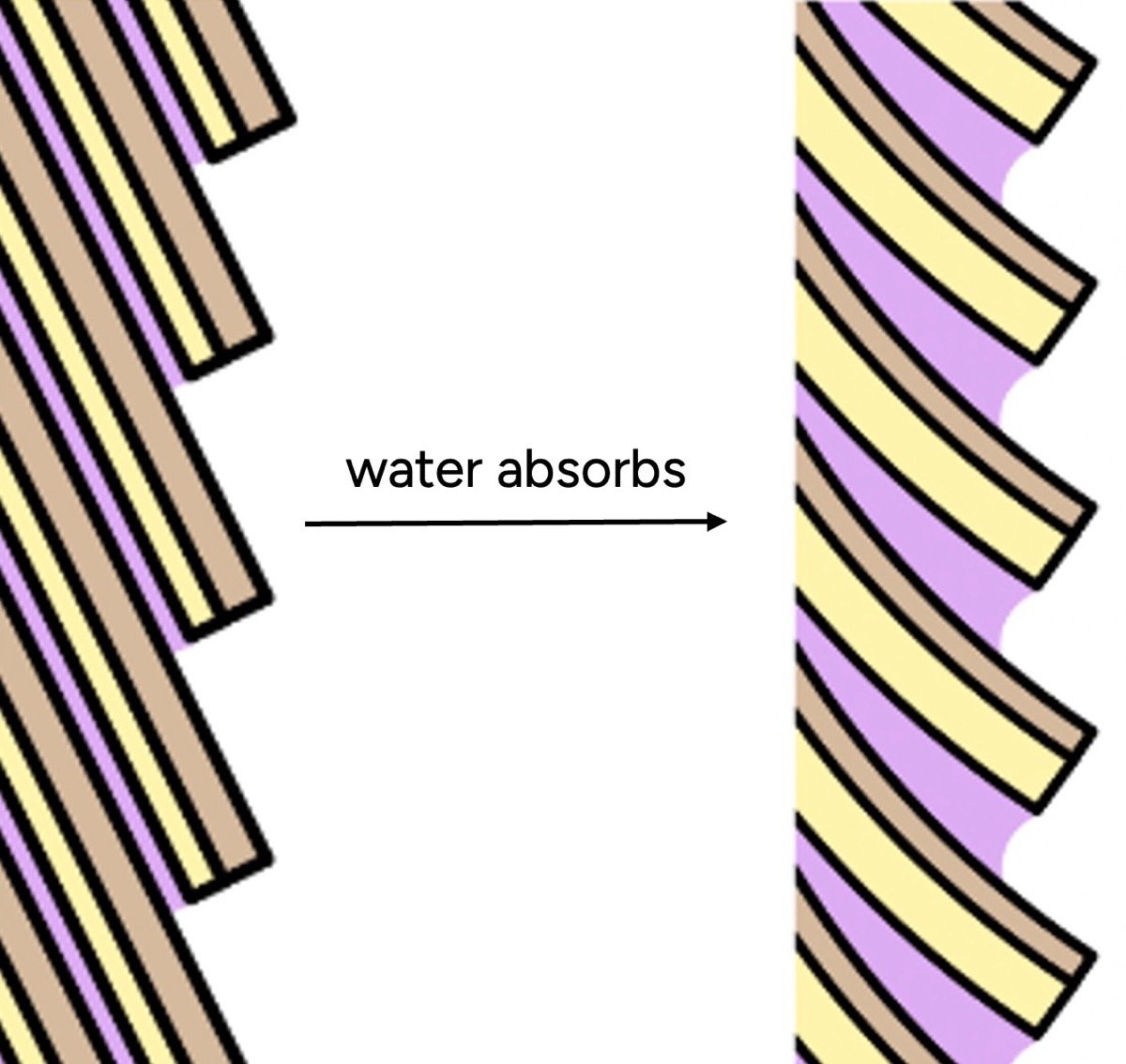
Beyond internal structure, water significantly affects the hair’s external feel. The outermost layer, the cuticle, is comprised of overlapping scales. In dry conditions, these scales lie relatively flat, contributing to a smooth surface. When hair absorbs water, however, the cuticle scales swell. Crucially, this swelling is uneven: the underside of each scale (endocuticle) and the intercellular glue (cell membrane complex) absorb significantly more water than the outer surface. This differential swelling causes the scales to lift and buckle, creating a microscopically rough and bumpy surface.
Furthermore, water itself possesses adhesive properties and increases surface friction. This is evident when we moisten our fingers for a better grip. Consequently, a hair strand with higher water content presents a bumpier, grippier surface. Our tactile perception, conditioned by experiences with other materials, interprets this roughness as "dryness" or damage, whereas in most contexts, wet objects tend to feel smoother. This sensory mismatch is a primary reason why "hydrated" hair, in the colloquial sense, feels smooth and soft, even if it has a lower actual water content.
Decoding "Hydrating" Products: The Role of Conditioners
Given the scientific reality, how do products marketed as "hydrating" actually work? The answer lies predominantly in conditioning. These products typically contain ingredients like silicones, fatty alcohols, and other emollients that deposit a thin, oily film on the hair cuticle. This film serves several critical functions:
- Smoothing: It smooths down the lifted and buckled cuticle scales, creating a uniform, less bumpy surface.
- Protection: It forms a protective barrier, mimicking the hair’s natural F-layer (a hydrophobic lipid layer that is often lost through damage).
- Friction Reduction: By reducing surface roughness and stickiness, it significantly lowers inter-strand friction, making hair feel soft, slippery, and easier to manage.
Therefore, the perceived "hydration" from these products is primarily a conditioning effect that enhances the hair’s tactile and visual properties, rather than an increase in its internal water content.
The Ritual of Washing: Beyond Cleanliness to Reshaping
Many individuals observe that their hair feels markedly better after a full wash and condition compared to merely applying a leave-in product. This phenomenon is not solely about "hydration" but involves a complex interplay of physical and chemical processes:
-
Optimized Conditioning Ingredient Distribution: Water acts as an excellent solvent and spreading agent. During a wash, the abundant water allows conditioning ingredients to disperse more evenly and coat each hair strand more comprehensively than a concentrated leave-in product might.
-
Hair Shape Reset: Water is exceptionally effective at resetting the hair’s shape. This is evident in how washing eliminates heat styling or how humidity disrupts a carefully crafted hairstyle. The heat and water in washing, coupled with the action of shampoo (which reduces surface tension, allowing deeper water penetration), profoundly disrupt temporary bonds. This renders the hair highly pliable, allowing it to be effectively reshaped as it dries. It’s analogous to molding warm candle wax – the softer it is, the more readily it can be reshaped.
-
Enhanced Clump Formation for Curly Hair: For curly and coily textures, water plays an additional, crucial role in promoting parallel strand alignment. Surface tension causes water to coat the hair strands, making them "sticky." When wet strands come into contact, water facilitates their "zipping" together, forming well-defined curl clumps. These larger, aligned clumps can create the illusion of "hydration" when dry. Their increased surface area in contact with the hand allows for faster heat transfer, making them feel colder and smoother than disaggregated, unaligned hair. Our brains often interpret this sensation as "moisturized" or "healthy."
-
Microscopic Cuticle Reset: Water can also rectify microscopic structural irregularities. Rapid drying, especially with hot tools, can cause uneven shrinkage and buckling of cuticle scales. Similarly, mechanical stretching (even as little as 15%) can induce cuticle buckling. Soaking hair in water and allowing it to dry naturally can effectively "reset" these buckled scales, restoring a smoother surface. This effect is about water facilitating a structural reorganization, not merely increasing water content.

Finding the Equilibrium: The Ideal Hair Water Content
Just as with skin, there is an optimal range for hair’s water content. Too much water, as discussed, can lead to weakened bonds, cuticle swelling, and increased friction. Conversely, hair with too little water also suffers negative consequences:
- Increased Static Build-up: In very low humidity environments (e.g., airplane cabins), hair can accumulate static electricity more easily. Water molecules help to dissipate electrical charges, so their absence exacerbates static, leading to flyaways.
- Brittleness and Reduced Shock Absorbency: Severely dry hair, with an excess of strong temporary bonds, becomes brittle and less flexible. It cannot adequately absorb mechanical stress from brushing or styling, leading to microscopic cracks that eventually result in breakage. This phenomenon is readily observed with fingernails, which, being structurally similar to hair, are prone to uncontrolled cracking when clipped dry, but offer cleaner cuts when wet and flexible.
Therefore, the goal is not maximal "hydration" but rather maintaining hair within an ideal moisture equilibrium where it possesses optimal flexibility, strength, and smoothness.
Practical Implications for Haircare and Product Development
The scientific understanding of water-hair interactions has profound implications for both product formulation and consumer haircare routines:
- Focus on Conditioning, Not Just "Hydration": Product developers should emphasize the conditioning and protective benefits of their formulations rather than solely relying on the ambiguous term "hydration." Ingredients that smooth the cuticle, reduce friction, and provide a protective layer are key.
- Personalized Detangling Strategies: Consumers should adjust their detangling methods based on hair type. Straight hair generally benefits from dry detangling with a wide-tooth comb and a leave-in conditioner, while curly hair often thrives with wet detangling, copious conditioner, and specialized tools.
- Gentle Wet Hair Handling: Regardless of hair type, wet hair is weaker. Gentle handling, avoidance of aggressive brushing, and the liberal use of conditioners are crucial to minimize damage. The myth of "hygral fatigue" (damage from repeated wetting and drying) is largely unfounded when proper care is taken.
- Humidity Management: Understanding how humidity affects hair structure and feel can help in selecting appropriate styling products and techniques. Anti-frizz products, for instance, often work by creating a hydrophobic barrier that minimizes water absorption by the cuticle.
- The Role of Emollients and Occlusives: While humectants (like glycerin) can attract water, their efficacy is dependent on environmental humidity. Emollients (like oils and silicones) and occlusives (like heavier oils or butters) are often more critical for managing hair feel and appearance, by smoothing the cuticle and reducing water loss from the hair shaft.
In conclusion, water is neither an inherent savior nor a destroyer of hair. It is a powerful solvent and structural modulator, capable of both weakening internal bonds and facilitating reshaping and conditioning. The perception of "hydrated" hair is often a sensory illusion created by a smooth, low-friction surface, rather than an elevated internal water content. By embracing the actual science of hair and water, consumers can move beyond marketing myths to adopt more effective, scientifically informed haircare practices that truly promote the health and aesthetic appeal of their hair.