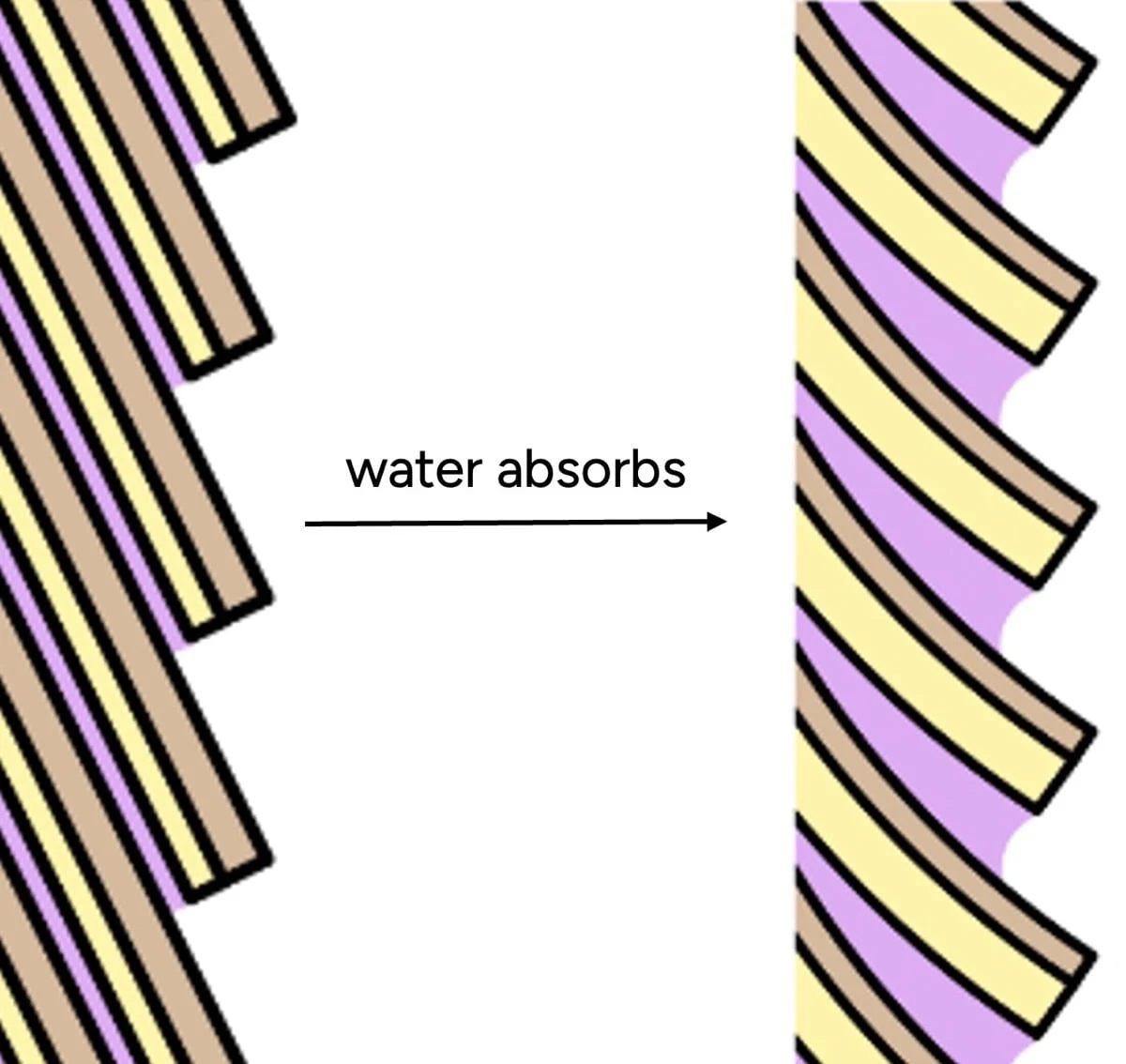
Understanding the intricate relationship between hair and water is paramount for effective hair care, yet it remains one of the most misunderstood topics, frequently obscured by marketing rhetoric and entrenched myths. While countless hair products promise "hydration" to achieve soft, smooth, and "healthy" locks, the scientific reality presents a far more complex picture, challenging both consumer perceptions and, at times, even expert consensus. This article delves into the molecular mechanics of water-hair interactions, debunking common misconceptions and providing a factual foundation for informed hair care practices.
The "Hydrated Hair" Paradox: Beyond Marketing Claims
The pervasive notion that "hydrated hair" is synonymous with high water content and superior health is a cornerstone of modern hair care marketing. However, scientific inquiry reveals a paradoxical truth: hair perceived as soft, smooth, and healthy often exhibits lower water content than hair that feels rough, dry, or damaged. This discrepancy highlights a fundamental disconnect between consumer language and scientific understanding, a challenge that even seasoned cosmetic scientists acknowledge. Industry conferences have seen experts advise against attempting to dismantle years of ingrained beliefs, fearing consumer alienation and competitive disadvantage. Despite this, a deeper dive into the science is essential for genuine hair health.
A notable study underscored this paradox, asking 50 individuals to evaluate hair samples with varying water content. A majority reported that hair with less water felt more moisturized, smoother, less tangled, and appeared less damaged. This counter-intuitive finding points to the critical role of the hair’s surface structure and sensory perception in defining "hydration" from a consumer standpoint, rather than the internal water levels.
The Molecular Architecture of Hair and Water’s Influence
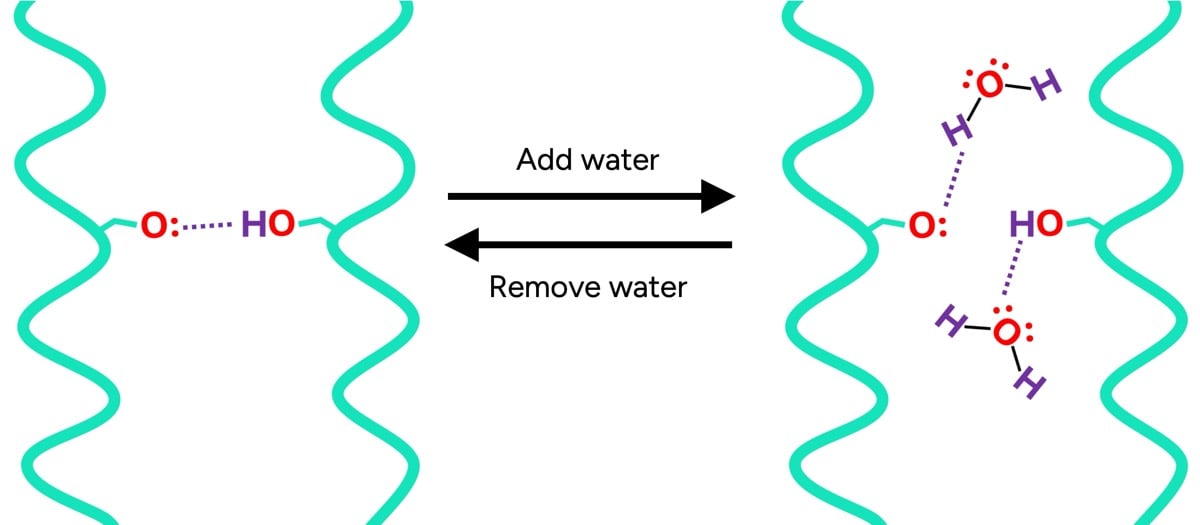
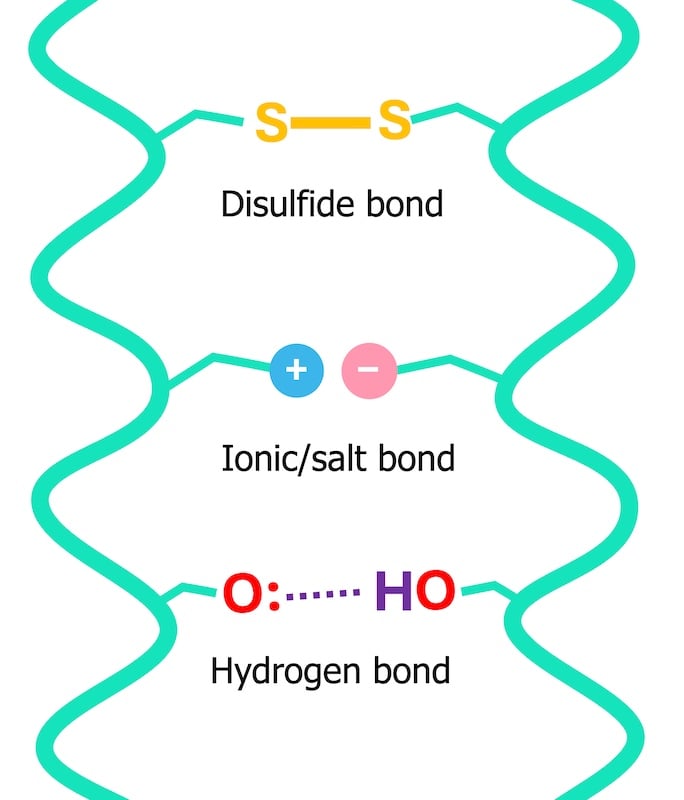
To truly grasp how water interacts with hair, one must first understand its fundamental composition. Hair is primarily composed of keratin proteins, which are held together by various types of chemical bonds, imparting strength and shape. These bonds fall into two main categories: permanent (covalent bonds like disulfide bonds) and temporary (hydrogen and ionic bonds). The temporary bonds, particularly hydrogen bonds, are highly susceptible to water.
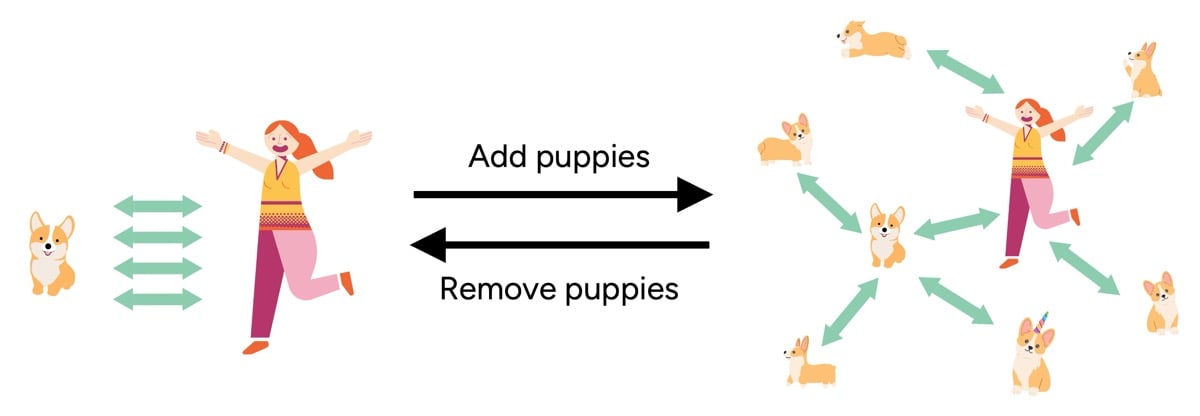
In dry hair, keratin proteins are extensively linked by temporary hydrogen bonds, acting like molecular "handshakes" that provide rigidity and stability. When water is introduced, its highly polar molecules compete with the keratin proteins for these hydrogen bonds. Water molecules effectively interpose themselves between the protein strands, disrupting the protein-protein bonds and forming new hydrogen bonds with individual keratin molecules. This molecular "dilution" means that wet hair has significantly fewer protein-to-protein bonds holding its structure together.
This phenomenon can be visualized through an analogy: imagine two individuals (representing keratin proteins) holding hands (hydrogen bonds). If a crowd of friendly puppies (water molecules) enters the scene, the individuals are likely to release each other’s hands to interact with the puppies. While they are still interacting, their direct bond with each other is weakened. Similarly, in wet hair, the keratin proteins become less "bonded" to each other, leading to a reduction in the hair’s internal structural integrity.
Structural Implications: Strength, Elasticity, and Damage
The disruption of temporary bonds by water has profound implications for hair’s mechanical properties:
- Weakening of Structure: Wet hair is inherently weaker than dry hair. The reduced number of protein-protein bonds makes it more vulnerable to mechanical stress.
- Increased Elasticity and Swelling: With fewer internal cross-links, wet hair becomes stretchier and more elastic. It also absorbs water and swells, increasing its diameter. This swelling can lead to the lifting of cuticle scales, further impacting its surface properties.
This dual effect explains why the act of detangling hair, particularly when wet, is a nuanced practice with varying outcomes depending on hair type.
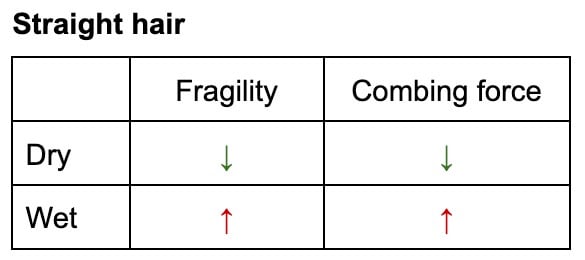
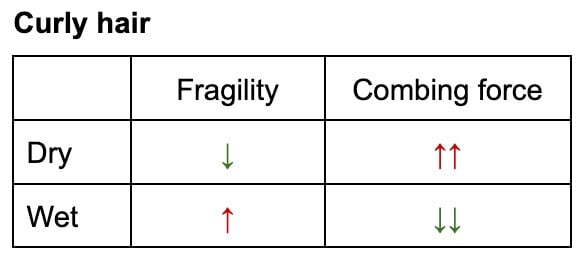
The Detangling Conundrum: Straight vs. Curly Hair
The recommendation for detangling hair—whether wet or dry—is not universal but highly dependent on hair’s natural texture:
- Straight Hair: For straight hair, wet detangling is generally more damaging. Although the hair is more elastic, its weakened state combined with increased friction between strands (due to water’s sticky nature) makes it highly susceptible to breakage. Combing through wet, straight hair often requires more force, leading to greater mechanical damage.
- Curly and Coily Hair: Conversely, for curly and coily hair, wet detangling is often less damaging. While the hair is still weaker when wet, the presence of water and, crucially, a good conditioner, helps to lubricate the strands and loosen tangles. The natural coiled structure, when saturated with water, becomes more pliable, reducing the resistance encountered during detangling. This allows for easier manipulation with less overall force and, consequently, less damage. Conditioners play a vital role here, providing a slippery coating that further reduces friction.
This "conundrum" is not limited to fully wet hair; even in humid environments, where hair absorbs ambient moisture, similar effects on strength and detangling resistance have been observed. The key takeaway is to minimize the force required for detangling, adapting the method to the specific hair type and ensuring ample lubrication with conditioning agents.
The Hair Surface: Cuticle Response and Sensory Perception
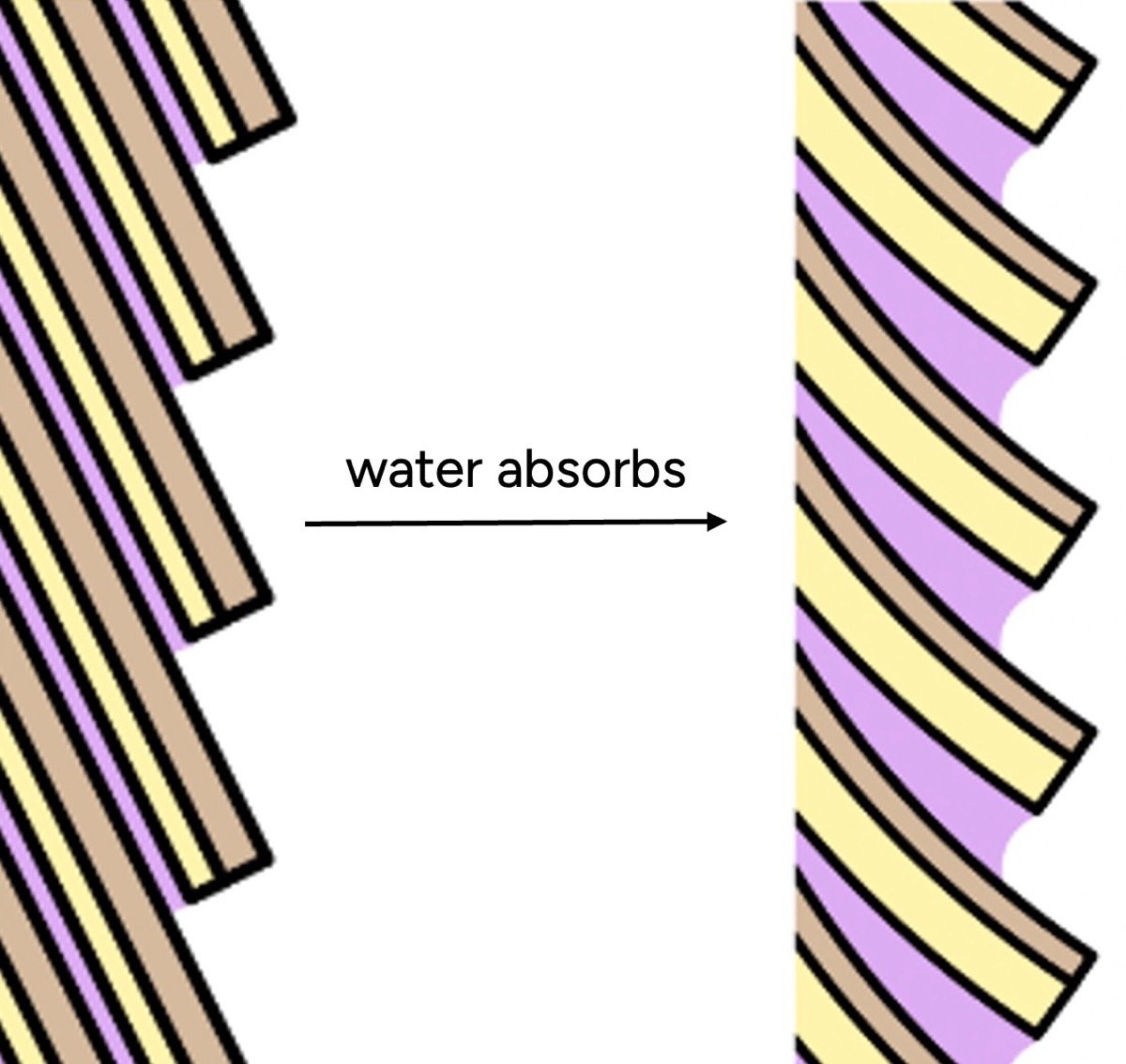
The feeling of "dryness" or "health" in hair is heavily influenced by the condition of its outermost layer, the cuticle. The cuticle consists of overlapping scales, similar to shingles on a roof.
- Cuticle Swelling: When hair absorbs water, these cuticle scales swell. Crucially, different parts of the cuticle swell unevenly. The underside of each scale (endocuticle) and the adhesive material between scales (cell membrane complex) absorb significantly more water than the outer, more robust layer. This differential swelling causes the scales to lift and protrude, creating a microscopically rough and bumpy surface.
- Increased Friction: Water itself is inherently sticky and increases friction. This property is commonly exploited when wetting fingers to get a better grip. On a microscopic level, this stickiness, combined with the raised cuticle scales, makes wet hair feel rougher and "grippier."
- Misinterpretation of "Dryness": Our tactile perception interprets this roughness as "dryness" or damage, largely because most other materials we encounter tend to feel smoother when wet. This sensory illusion reinforces the misconception that increased water content equates to damaged hair, when in fact, the rough texture is a direct consequence of water’s interaction with the cuticle.
The Mechanism of "Hydrating" Products
Given this scientific reality, how do products marketed as "hydrating" actually work? The answer lies not in increasing the hair’s internal water content, but primarily in conditioning its surface.
- Conditioning Action: Most "hydrating" products are, at their core, conditioners. They contain oily ingredients such as silicones, fatty alcohols, and emollients that deposit a thin, protective film on the cuticle. This film serves to smooth down the lifted cuticle scales, reduce microscopic bumps, and decrease surface friction. The result is hair that feels soft, smooth, and appears shiny – sensations that consumers associate with "hydration."
- Mimicking the F-Layer: Undamaged hair naturally possesses a protective outer layer known as the F-layer, which is a covalently bonded fatty acid layer. This layer is hydrophobic, repelling water and contributing to the hair’s natural smoothness and luster. Damage (e.g., from heat styling, chemical treatments, harsh brushing) can degrade or remove the F-layer. Conditioners effectively attempt to replicate the protective and smoothing functions of this natural oily barrier.
The Ritual of Washing: More Than Just Cleanliness
Many consumers observe that hair feels significantly better after a full wash and condition compared to simply applying a leave-in product. This phenomenon involves several scientific principles, none of which are solely about "adding water" to the hair:
- Optimized Ingredient Distribution: Water acts as an excellent solvent and spreading agent. During a rinse-off conditioning treatment, the abundance of water ensures that conditioning ingredients are dispersed more evenly and thoroughly across each hair strand, leading to a more uniform and effective coating.
- Resetting Hair Shape: Water is remarkably effective at resetting the hair’s shape. This is evident when washing away heat styling or observing how humidity disrupts a carefully styled coiffure. The mechanism involves the temporary bonds within the hair’s cortex. Heat styling or environmental factors create new temporary bonds that hold hair in a desired shape. When water penetrates the hair, especially with the aid of shampoo (which reduces surface tension), it breaks these temporary bonds. This allows the hair’s permanent bonds to guide it back to its natural, relaxed state. This "floppy" state makes hair highly pliable and receptive to new styling.
- Enhanced Curl Clumping (for curly/coily hair): For those with curly and coily textures, water plays an additional crucial role in facilitating curl formation. The surface tension of water causes wet hair strands to adhere to one another, aligning them into distinct, well-defined clumps. This process, known as self-assembly, creates larger, more uniform curl patterns. These larger clumps, when dry, present a smoother, more aligned surface area to the touch. This can lead to a sensory perception of being "hydrated" because smooth, aligned surfaces transfer heat more efficiently and thus feel cooler and smoother than disordered hair, akin to how satin feels cooler than rough cotton.
- Microscopic Cuticle Reset: Rapid or uneven drying, particularly with high heat, can cause cuticle scales to buckle or warp due to differential shrinkage. Mechanical stress, even moderate stretching (around 15%), can also contribute to this buckling. Soaking hair in water and allowing it to dry naturally and gently can help "reset" these buckled scales, allowing them to lie flatter and contribute to a smoother overall surface. This is a shape-reforming effect, not simply about water retention.
The Ideal Water Content: A "Goldilocks Zone"
Just as with skin, hair thrives within an optimal range of water content – a "goldilocks zone" where it is neither too wet nor too dry.
- Too Much Water: While the concept of "hygral fatigue" (damage from repeated wetting and drying) is largely considered a myth, consistently saturated hair is indeed weaker, rougher, and more prone to mechanical damage during manipulation.
- Too Little Water: Conversely, hair that is excessively dry suffers from:
- Increased Static Electricity: In very low humidity environments, hair readily builds up static charge, leading to flyaways. Water molecules act as tiny conductors, helping to dissipate these charges.
- Brittleness and Reduced Shock Absorption: Without a sufficient, optimal level of internal moisture, the hair’s numerous protein-protein bonds make it rigid and inflexible. This rigidity prevents it from effectively absorbing mechanical shocks (e.g., from brushing or styling), leading to the formation and propagation of microscopic cracks that ultimately result in breakage. This phenomenon is readily observable in fingernails, which share similar protein structures with hair; dry nails crack and split more readily than wet, flexible nails when clipped.
Strategies for Optimizing Hair’s Water Content and Feel
Achieving the ideal state for hair involves managing its interaction with water and conditioning its surface, rather than solely focusing on "adding hydration."
- Conditioners and Oils: These products create a protective hydrophobic barrier on the hair surface, which helps to regulate the rate of water exchange with the environment. They prevent excessive water absorption that leads to cuticle swelling and weakening, and they also minimize water loss from the hair’s interior, maintaining a balanced moisture level.
- Humectants: Ingredients like glycerin, hyaluronic acid, or panthenol can attract and hold a beneficial amount of moisture from the air, helping to keep hair pliable without over-saturating it. Their effectiveness, however, is highly dependent on ambient humidity.
- Gentle Handling: Regardless of water content, gentle handling is crucial. Using wide-tooth combs, starting detangling from the ends, and applying ample conditioner significantly reduces mechanical stress and prevents breakage, particularly when hair is in its more vulnerable wet state.
- Heat Protection: Minimizing high heat styling, or using heat protectants, helps preserve the F-layer and the structural integrity of the cuticle, allowing hair to naturally regulate its moisture more effectively.
Implications for Consumers and the Hair Care Industry
The scientific understanding of hair and water has significant implications. For consumers, it empowers them to look beyond misleading marketing terms and make informed product choices based on actual hair needs. Instead of chasing "hydration," the focus should shift to effective conditioning, surface smoothing, and maintaining the hair’s optimal mechanical properties. Products that strengthen bonds (e.g., bond-repairing treatments), provide effective conditioning, and manage moisture exchange are key to genuinely healthy hair.
For the hair care industry, this knowledge presents a continuous challenge to balance scientific accuracy with market appeal. There’s a growing imperative for transparency in product claims and a focus on educating consumers about the true mechanisms behind healthy-feeling hair. Future innovations will likely center on sophisticated formulations that optimize the hair’s natural barriers, reinforce its internal structure, and control its interactions with environmental moisture, moving away from simplistic notions of "hydration."
In conclusion, water is neither an unequivocal boon nor a villain for hair. It is a powerful, dynamic tool that, when understood and managed correctly, can be leveraged to soften hair’s structure for styling, distribute beneficial ingredients, and reset its natural form. The goal is not simply to "hydrate" but to manage water’s interaction with hair to achieve optimal strength, elasticity, and a smooth, healthy appearance. Gentle care, particularly when hair is wet and at its most vulnerable, combined with effective conditioning, remains the cornerstone of true hair health, regardless of what product labels claim.
References

Wong, M. (2026, January 28). Hair, hydration and water: the real science. Lab Muffin Beauty Science. Accessed March 11, 2026. https://labmuffin.com/hair-hydration-and-water-the-real-science/
Epps, J., & Wolfram, L. J. (1983). Letter to the editor. J Soc Cosmet Chem, 34, 213-214.
Evans, T. (2013). Consumer vs. scientific language: relating in vivo to in vitro. Cosmetics & Toiletries, 128(5), 300-304.
Gamez-Garcia, M. (2021). Moisture in the cuticle sheath: effects on hair mechanical and cosmetic properties. J Cosmet Sci, 72, 687-696.
Kamath, Y. K., & Hornby, S. B. (1985). Effect of chemical and humectant treatments on the mechanical and fractographic behavior of Negroid hair. J Soc Cosmet Chem, 36, 39-52.
Kaushik, V., Chogale, R., & Mhaskar, S. (2021). Single hair fiber assessment techniques to discriminate between mineral oil and coconut oil effect on hair physical properties. J Cosmet Dermatol, 20(4), 1306-1317. doi:10.1111/jocd.13724
Robbins, C. R. (2012). Chemical and physical behavior of human hair (5th ed.). Springer Berlin Heidelberg.
Affiliate Disclosure: I receive a small commission for purchases made via affiliate links.